Animal health rules for non-EU countries: 2026 update
- Animal health
Summary
The European Commission has informed the World Trade Organization Sanitary and Phytosanitary Measures (WTO SPS) Committee that it is updating the animal health requirements for non-EU countries exporting to the European Union (EU) (G/SPS/N/EU/920).
The changes include:
- removal of gelatine, collagen, and highly refined products from the scope of animal health requirements – animal health rules will no longer apply to these products
- update of identification methods for certain animals
- extended application of specific requirements in relation to horse diseases
- stricter rules on raw meat from countries with foot and mouth disease
- update of risk mitigating treatments for dairy and eggs
- clarification of risk mitigating treatments for dairy and egg ingredients in composite products
- review of information in vaccination programmes against highly pathogenic avian influenza (HPAI)
- update of the list of EU Member State measures on aquatic species susceptible to diseases
- identification of vector species for the protozoan parasite Mikrocytos mackini.
Stakeholders in non-EU countries that are members of the WTO can submit comments on this proposal via their competent authorities until 18 April 2026.
European Commission proposes updated animal health rules for non-EU countries
Draft Commission Delegated Regulation (EU) amending and correcting Delegated Regulation (EU) 2020/692 supplementing Regulation (EU) 2016/429 of the European Parliament and of the Council as regards rules for entry into the Union, and the movement and handling after entry of consignments of certain animals, germinal products and products of animal origin
Draft Annex
Update
The European Commission has informed the World Trade Organization Sanitary and Phytosanitary Measures (WTO SPS) Committee that it is updating the animal health requirements for non-EU countries exporting to the European Union (EU) (G/SPS/N/EU/920).
The changes include:
- removal of gelatine, collagen, and highly refined products from the scope of animal health requirements – animal health rules will no longer apply to these products
- update of identification methods for certain animals
- extended application of specific requirements in relation to horse diseases
- stricter rules on raw meat from countries with foot and mouth disease
- update of risk mitigating treatments for dairy and eggs
- clarification of risk mitigating treatments for dairy and egg ingredients in composite products
- review of information in vaccination programmes against highly pathogenic avian influenza (HPAI)
- update of the list of EU Member State measures on aquatic species susceptible to diseases
- identification of vector species for the protozoan parasite Mikrocytos mackini.
Stakeholders in non-EU countries that are members of the WTO can submit comments on this proposal via their competent authorities until 18 April 2026.
Impacted Products
Dairy, shelf-stable products, milk, eggs and egg products, aquatic species
What is changing?
The European Commission proposes the following changes to animal health rules.
Gelatine, collagen, and highly refined products
As production of these products eliminates any risk to animal or public health, they will no longer have to comply with the European Union’s animal health rules (they are removed from the scope of Regulation 2020/692).
Identification of certain animals
The identification of certain animals must conform with ISO standards. As the ISO standards have been updated, references to the standards must also be updated in Regulation 2020/692 in relation to:
- ungulates – conventional or electronic ear tag (Art. 21)
- ratites – neck-tag or injectable transponder (Art. 43)
- captive birds – closed-ring or injectable transponder (Art. 53).
Extended application of requirements in relation to horse diseases
The specific requirements that apply to exported horses when horse diseases have been identified will be amended. They are to be extended to cases where African horse sickness, Venezuelan equine encephalomyelitis, or infection with glanders (Burkholderia mallei), dourine and surra (Trypanosoma) have been reported during certain periods (set out in Regulation 2020/692, Annex XI, 2.1). It is also proposed to remove these specific conditions for non-EU countries where these diseases have never been reported or have been absent for 24 months (36 months in the case of glanders).
Stricter rules on raw meat from countries with foot and mouth disease
To mitigate the risks of spreading foot and mouth disease, raw meat used for the production of meat products to be exported to the EU should not come from any country or territory in which cattle are vaccinated against this disease (Arts. 148 and 149).
Raw material to produce dairy or egg products
The proposal clarifies that:
- dairy products subject to a risk mitigating treatment can be produced from raw milk and dairy products (there is currently only a reference to “milk”, Art. 157).
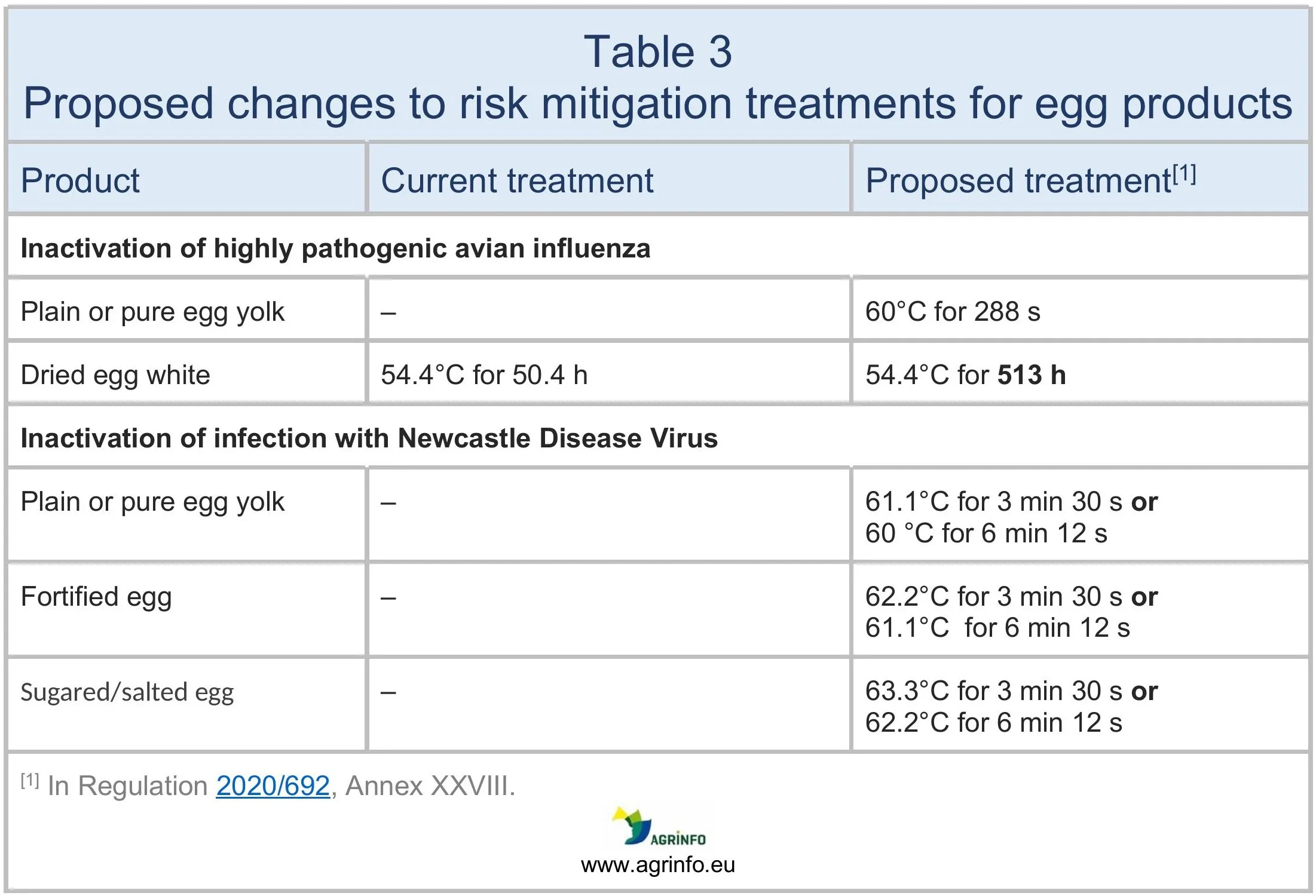
- egg products can enter the EU from authorised and listed non-EU countries not only if they have disease surveillance programmes in place for HPAI, but also if the egg products have been submitted to risk mitigation treatments that inactivate HPAI virus and Newcastle disease virus (Art. 160)
Composite products
The proposal clarifies the following (revised Art. 163).
- The general animal health requirements that apply to animal products (Regulation 2020/692, Part IV, Title 1) also apply to composite products. This includes the application of rules on risk mitigating treatments for certain products from certain non-EU countries or territories.
- The requirements for countries authorised to produce shelf-stable composite products in relation to the risk mitigating treatments for imported dairy ingredients (see Table 1).
- Dairy and egg ingredients that have undergone sterilisation or ultra-high-temperature (UHT) processing (see Regulation 2020/692, Annex XXVII, column B for dairy; Annex XXVIII, points 1 and 2 for eggs) can be used by manufacturers of composite products in countries not specifically listed for those products if those countries are nevertheless listed for meat products, dairy products, or egg products
Risk mitigating treatments for dairy and egg products
These treatments have been updated (see Tables 2 and 3).
Information in vaccination programme against HPAI
The minimum information to be included in vaccination programmes against HPAI is reviewed, revising Regulation 2020/692, Annex XIII to align with Regulation 2023/361, Annex III.
Export of horses to the EU
The proposal clarifies that only animals free from horse sickness and that have not been vaccinated against this disease can be exported to the EU (revision of Regulation 2020/692, Annex IV).
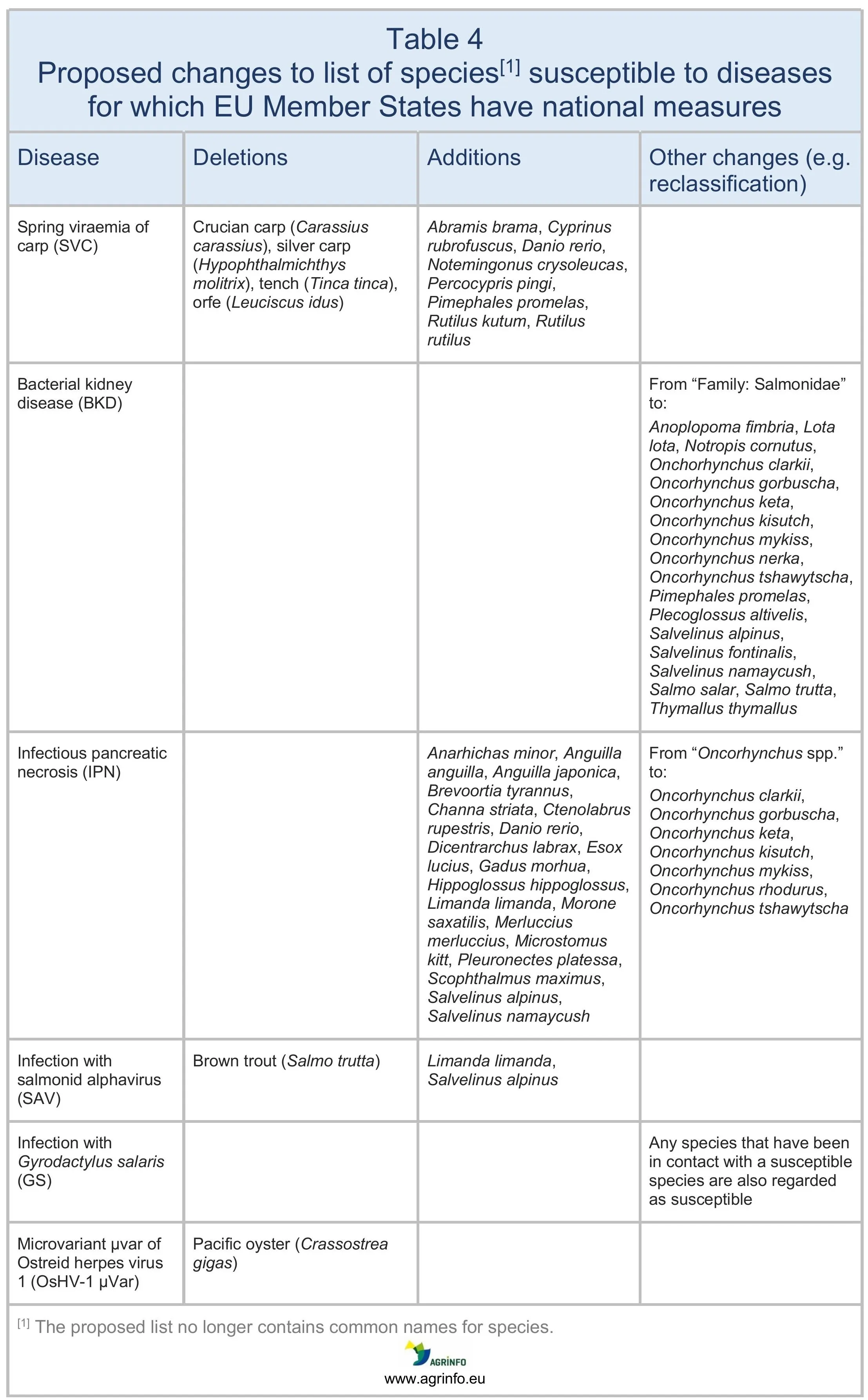
EU Member State measures on aquatic species susceptible to diseases
An update of this list is proposed (see Table 4) (revision of Regulation 2020/692, Annex XXIX).
Vector species identified for Mikrocytos mackini
The eastern oyster (Crassostrea virginica) is listed as a vector for the protozoan parasite Mikrocytos mackini (added to Regulation 2020/692, Annex XXX; no vectors were previously identified).
Why?
New scientific knowledge and the experience gained in applying Regulation 2022/692 have shown the need to refine several provisions related to the scope, application of specific conditions, animal identification, meat, dairy and egg products, composite products, vaccination programmes against highly pathogenic avian influenza, and aquatic animals.
Timeline
The date of application is yet to be determined.
What are the major implications for exporting countries?
Producers of dairy and egg products should pay attention to the proposed changes in risk mitigating treatments.
Recommended Actions
Competent authorities of countries that are members of the WTO can submit comments on the EU’s proposal by emailing the EU SPS Enquiry Point until 18 April 2026.
Background
Exports of animals and animal products to the EU are subject to animal health requirements to prevent the spread of diseases.
For non-EU countries, the key requirements are in Delegated Regulation 2020/692 (see Animal health requirements for non-EU countries exporting to the EU – explained).
Resources
Commission Delegated Regulation (EU) 2020/692 as regards rules for entry into the Union, and the movement and handling after entry of consignments of certain animals, germinal products and products of animal origin
Sources
Disclaimer: Under no circumstances shall COLEAD be liable for any loss, damage, liability or expense incurred or suffered that is claimed to have resulted from the use of information available on this website or any link to external sites. The use of the website is at the user’s sole risk and responsibility. This information platform was created and maintained with the financial support of the European Union. Its contents do not, however, reflect the views of the European Union.
European Commission proposes updated animal health rules for non-EU countries
Draft Commission Delegated Regulation as regards rules for entry into the Union, and the movement and handling after entry of consignments of certain animals, germinal products and products of animal origin
Draft Annex
What is changing and why?
Based on new scientific knowledge and the experience gained in applying Regulation 2020/692, the European Commission proposes the following changes.
- Removal of gelatine, collagen, and highly refined products from the scope of animal health requirements (animal health rules will no longer apply to these products).
- Update of the identification methods for ungulates (conventional or electronic ear tag), ratites (neck-tag or injectable transponder), and captive birds (closed-ring or injectable transponder) to align with ISO standards.
- Extending the application of specific animal health requirements in relation to horse diseases to include cases where African horse sickness, Venezuelan equine encephalomyelitis, or infection with glanders, dourine or surra have been reported during certain periods; and removing these requirements where these diseases have never been reported or have been absent for 24 months (36 months in the case of glanders).
- Setting stricter rules regarding foot and mouth disease: raw meat used for the production of meat products to be exported to the EU must not come from any country or territory in which cattle are vaccinated against this disease.
- Allowing the production of dairy and egg products from products that have been submitted to risk mitigation treatments.
- For shelf-stable composite products, clarifying the rules authorising countries to produce both the dairy ingredients for these products, and the composite products themselves. The requirements depend on the risk mitigating treatment applied to the dairy ingredient (Table 1).
- Updating the risk mitigating treatments for dairy and egg products (Tables 2 and 3).
- Reviewing the information in vaccination programmes against highly pathogenic avian influenza (HPAI).
- Updating the list of aquatic species susceptible to diseases for which EU Member States have national measures (Table 4).
- Adding eastern oyster (Crassostrea virginica) as a vector for infection with the protozoan parasite Mikrocytos mackini (no vectors were previously identified).
Actions
Competent authorities of countries that are members of the World Trade Organization (WTO) can submit comments on the EU’s proposal by emailing the EU SPS Enquiry Point until 18 April 2026.
Timeline
The date of application is yet to be determined.
Tables & Figures
Disclaimer: Under no circumstances shall COLEAD be liable for any loss, damage, liability or expense incurred or suffered that is claimed to have resulted from the use of information available on this website or any link to external sites. The use of the website is at the user’s sole risk and responsibility. This information platform was created and maintained with the financial support of the European Union. Its contents do not, however, reflect the views of the European Union.