Feed additives: January–February 2026 authorisations, reauthorisations, and changes
- Feed additives
- Feed safety
Summary
Overview of the latest European Union (EU) authorisations and reauthorisations of feed additives and their use in animal nutrition in target animals, including changes to existing authorisations.
EU authorises and reauthorises certain feed additives
Commission Implementing Regulations (EU) 2026/85, 2026/93, 2026/96, 2026/98, 2026/103, 2026/119, 2026/154, 2026/164, 2026/167, 2026/168, 2026/171, 2026/178, 2026/180, 2026/348, 2026/352, 2026/353, 2026/356, 2026/402, 2026/404, 2026/460
Update
Overview of the latest European Union (EU) authorisations and reauthorisations of feed additives and their use in animal nutrition in target animals, including changes to existing authorisations.
Impacted Products
Feed additives, prepared fodder
What is changing?
Authorisations
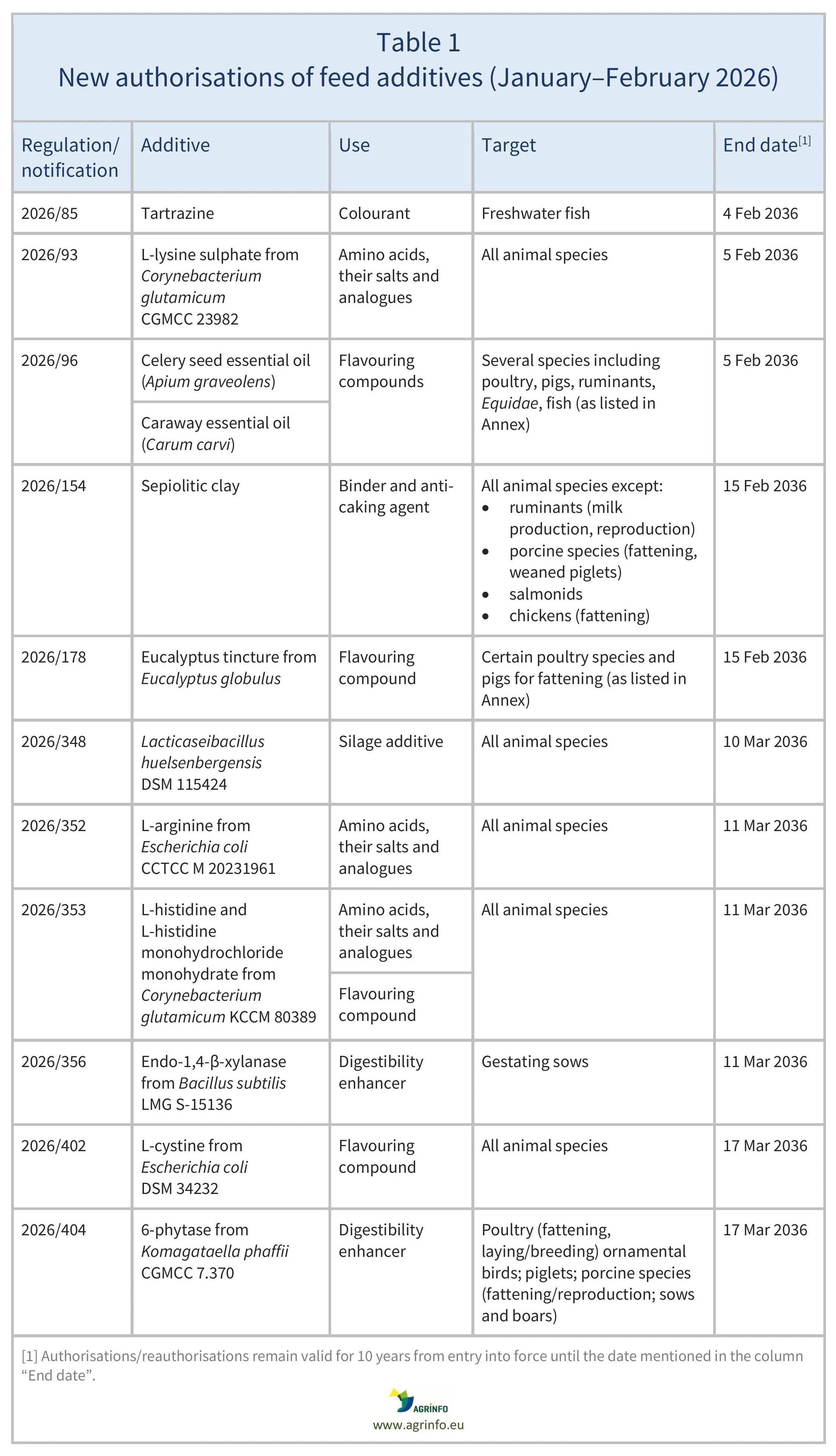
In January–February 2026, the EU authorised the feed additives listed in Table 1, based on opinions published by the European Food Safety Authority (EFSA) [see Resources 3, 5–7, 9, 12, 17, 19–24]. The conditions of use are described in the respective Regulations.
Reauthorisations
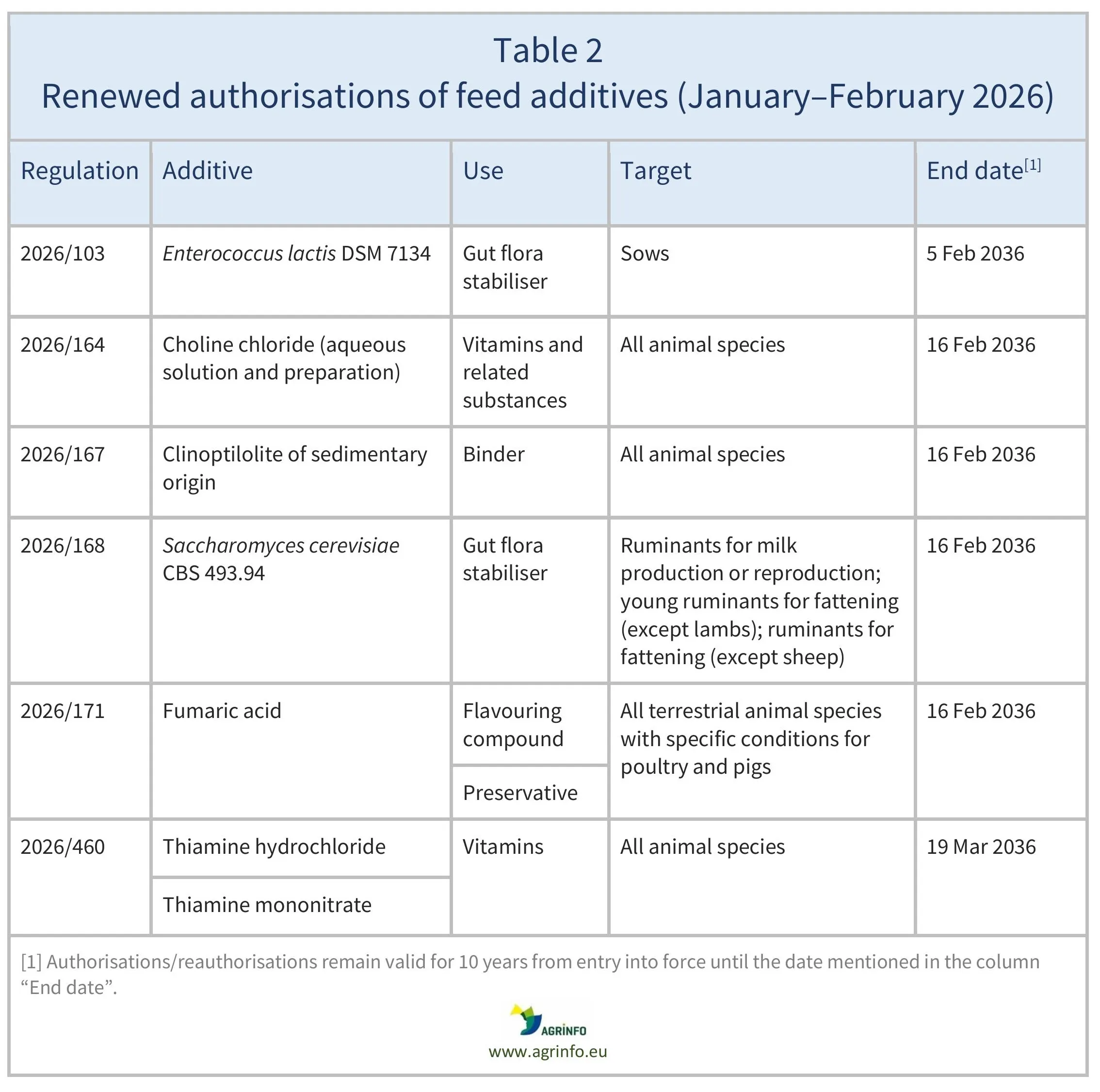
In January–February 2026, the EU reauthorised the feed additives listed in Table 2, based on opinions published by EFSA [see Resources 8, 11, 13–16, 18]. The conditions of use are described in the respective Regulations.
Refusal
Regulation 2026/119 refuses authorisation of a preparation of carvacrol as a feed additive belonging to the category of zootechnical additives for weaned piglets.
Changes
Regulation 2026/98 updates the name of the authorisation holder for a preparation of 3-nitrooxypropanol to dsm-firmenich.
Regulation 2026/154 extends authorisation for the use of sepiolitic clay as a binder and anti-caking agent to include additional species, and increases the maximum content to 20,000 mg/kg feed for salmonids and chickens for fattening.
Regulation 2026/180 changes the name of the authorisation holder for the feed additives serine protease, endo-1,4-beta-xylanase, endo-1,3(4)-beta-glucanase and endo-1,4-beta-glucanase, endo-1,3(4)-beta-glucanase, endo-1,4-beta-xylanase, 6-phytase, protease, and muramidase.
The European Commission has informed the World Trade Organization Sanitary and Phytosanitary Measures (WTO SPS) Committee that it intends to amend Regulation 2020/1097 authorising lutein-rich and lutein/zeaxanthin extracts from Tagetes erecta as feed additives for poultry (except turkeys) for fattening and laying, and Regulation 2025/1928 authorising a preparation of lutein-rich extract of T. erecta as a feed additive for turkeys for fattening (G/SPS/N/EU/921).
Why?
Authorisations
Applications for the above authorisations and reauthorisations were submitted and considered by the Reference Laboratory set up by the Feed Additives Regulation 1831/2003.
Refusal
Regulation 2026/119 refuses authorisation of a preparation of carvacrol because the applicant has not adequately demonstrated its efficacy as a feed additive for weaned piglets [see Resources 2, 4].
Changes
Amendments to Regulations 2020/1097 and 2025/1928 are considered necessary due to the similarities in the manufacturing process and composition of the additives authorised [see Resources 1, 10].
Timeline
These authorisations and reauthorisations remain valid until the end dates listed in Tables 1 and 2.
What are the major implications for exporting countries?
With these authorisations, more feed additives will be available on the market. Authorisations and renewals are valid for 10 years. The use of all preparations and substances specified as feed additives must comply with the provisions of use specified in the Annex to each Regulation.
Recommended Actions
Non-EU countries producing feed additives, compound feed, and feed materials for export to the EU are recommended to check the status of the feed additives in the EU Feed Additives register.
To be able to filter and to see more information, it is advised to download the register in Excel format (see the Food and Feed Information Portal webpage).
Background
The procedure for authorising the placing on the market and use of feed additives is set out in Regulation (EC) 1831/2003. For the latest updates on feed additives, see the EU Feed Additives register.
Resources
- EFSA (2019) Safety and efficacy of lutein and lutein/zeaxanthin extracts from Tagetes erecta for poultry for fattening and laying (except turkeys). EFSA Journal, 17(5): e5698.
- EFSA (2020) Safety and efficacy of Nimicoat® (carvacrol) as a zootechnical additive for weaned piglets. EFSA Journal, 18: 6070.
- EFSA (2022) Safety and efficacy of 6-phytase produced by Komagataella phaffii CGMCC 7.370 for pigs and avian species (Victory Enzymes GmbH). EFSA Journal, 20: 7701.
- EFSA (2024) Efficacy of a feed additive consisting of carvacrol (Nimicoat®) for weaned piglets (Techna France Nutrition). EFSA Journal, 22: e8639.
- EFSA (2024) Safety and efficacy of a feed additive consisting of a tincture derived from the leaves of Eucalyptus globulus Labill. (eucalyptus tincture) for all animal species (FEFANA asbl). EFSA Journal, 22(5): e8801.
- EFSA (2024) Safety and efficacy of a feed additive consisting of an essential oil obtained from the fruit of Carum carvi L. (caraway oil) for all animal species (FEFANA asbl). EFSA Journal, 22(7): e8906.
- EFSA (2024) Safety and efficacy of a feed additive consisting of an essential oil obtained from the fruit of Apium graveolens L. (celery seed oil) for all animal species (FEFANA asbl). EFSA Journal, 22(7): e8907.
- EFSA (2024) Safety and efficacy of a feed additive consisting of fumaric acid for all animal species for the renewal of its authorisation and extension of use (Life SUPPLIES NV). EFSA Journal, 22: e9019.
- EFSA (2024) Safety and efficacy of a feed additive consisting of tartrazine for its use in baits for freshwater fish (GIFAP). EFSA Journal, 22(10): e9021.
- EFSA (2024) Safety and efficacy of a feed additive consisting of lutein-rich extract of Tagetes erecta L. for turkeys for fattening (EW Nutrition). EFSA Journal, 22(10): e9027.
- EFSA (2025) Assessment of the feed additive consisting of choline chloride for all animal species for the renewal of its authorisation (Andrés Pintaluba S.A., Taminco B.V., Balchem Italia S.r.l.). EFSA Journal, 23: e9264.
- EFSA (2025) Safety and efficacy of a feed additive consisting of L-lysine sulfate produced with Corynebacterium glutamicum CGMCC 23982 for all animal species (Eppen Europa SAS). EFSA Journal, 23(4): e9346.
- EFSA (2025) Assessment of the feed additives thiamine hydrochloride (3a820) and thiamine mononitrate for all animal species for the renewal of their authorisation (DSM Nutritional Products Ltd. and Orffa Additives BV). EFSA Journal, 23: e9347.
- EFSA (2025) Assessment of the feed additive consisting of Enterococcus lactis DSM 7134 (Bonvital®) for sows for the renewal of its authorisation (Lactosan GmbH & Co.KG). EFSA Journal, 23: e9353.
- EFSA (2025) Assessment of the feed additive consisting of Saccharomyces cerevisiae CBS 493.94 (Yea-Sacc®) for dairy cows, minor dairy ruminant species, cattle for fattening and minor ruminant species for fattening for the renewal of its authorisation (Alltech Ireland). EFSA Journal, 23: e9354.
- EFSA (2025) Safety and efficacy of the feed additive consisting of clinoptilolite of sedimentary origin for all animal species for the renewal of its authorisation (ZEOCEM, a.s.). EFSA Journal, 23: e9364.
- EFSA (2025) Safety of sepiolitic clay for all animal species (MYTA S.A.). EFSA Journal, 23: e9365.
- EFSA (2025) Assessment of the feed additives thiamine hydrochloride (3a820) and thiamine mononitrate (3a821) (vitamin B1) for all animal species for the renewal of their authorisation (Kaesler Nutrition GmbH). EFSA Journal, 23: e9405.
- EFSA (2025) Safety and efficacy of Lacticaseibacillus huelsenbergensis DSM 115424 as a silage additive (Lactosan GmbH & Co. KG). EFSA Journal, 23: e9458.
- EFSA (2025) Efficacy of a feed additive consisting of tartrazine for its use in baits for freshwater fish (GIFAP). EFSA Journal, 23(6): e9461.
- EFSA (2025) Safety and efficacy of L-histidine additives produced with Corynebacterium glutamicum KCCM 80389 (CJ Europe GmbH). EFSA Journal, 23: e9535.
- EFSA (2025) Safety and efficacy of endo-1,4-β-xylanase produced by Bacillus subtilis LMG S-15136 for gestating sows (Puratos NV). EFSA Journal, 23: e9552.
- EFSA (2025) Safety and efficacy of L-arginine produced with Escherichia coli CCTCC M 20231961 for all animal species (Kempex Holland B.V.). EFSA Journal, 23: e9609.
- EFSA (2025) Safety and efficacy of L-cystine produced with Escherichia coli DSM 34232 for all animal species (Wacker Chemie AG). EFSA Journal, 23: e9688.
Commission Implementing Regulation (EU) 2020/1097 concerning the authorisation of lutein-rich and lutein/zeaxanthin extracts from Tagetes erecta as feed additives for poultry (except turkeys) for fattening and laying and for minor poultry species for fattening and laying
Commission Implementing Regulation (EU) 2025/1928 concerning the authorisation of a preparation of lutein-rich extract of Tagetes erecta L. as a feed additive for turkeys for fattening
Regulation (EC) No 1831/2003 on additives for use in animal nutrition
Sources
Disclaimer: Under no circumstances shall COLEAD be liable for any loss, damage, liability or expense incurred or suffered that is claimed to have resulted from the use of information available on this website or any link to external sites. The use of the website is at the user’s sole risk and responsibility. This information platform was created and maintained with the financial support of the European Union. Its contents do not, however, reflect the views of the European Union.
EU authorises and reauthorises certain feed additives
Commission Implementing Regulations (EU) 2026/85, 2026/93, 2026/96, 2026/98, 2026/103, 2026/119, 2026/154, 2026/164, 2026/167, 2026/168, 2026/171, 2026/178, 2026/180, 2026/348, 2026/352, 2026/353, 2026/356, 2026/402, 2026/404, 2026/460
What is changing and why?
Authorisations and reauthorisations
In January and February 2026, the EU authorised and reauthorised the feed additives listed in Tables 1 and 2. These authorisations are based on opinions published by the European Food Safety Authority (EFSA). The conditions of use are described in the respective Regulations.
Refusal
Regulation 2026/119 refuses authorisation of a preparation of carvacrol because the applicant has not adequately demonstrated its efficacy as a feed additive for weaned piglets.
In addition, changes to some existing Regulations regarding feed additives are listed in the AGRINFO Full report.
Actions
Non-EU countries producing feed additives, compound feed, and feed materials for export to the EU are recommended to check the status of the feed additives in the EU Feed Additives register.
To be able to filter and to see more information, it is advised to download the register in Excel format (see the Food and Feed Information Portal webpage).
Timeline
These authorisations and reauthorisations remain valid until the end dates listed in Tables 1 and 2.
Tables & Figures
Disclaimer: Under no circumstances shall COLEAD be liable for any loss, damage, liability or expense incurred or suffered that is claimed to have resulted from the use of information available on this website or any link to external sites. The use of the website is at the user’s sole risk and responsibility. This information platform was created and maintained with the financial support of the European Union. Its contents do not, however, reflect the views of the European Union.