Flavourings: 3-[3-(2-isopropyl-5-methyl-cyclohexyl)-ureido]-butyric acid ethyl ester, and hesperetin dihydrochalcone
- Food additives
- Food safety
Summary
The European Union (EU) has added two substances – 3-[3-(2-isopropyl-5-methyl-cyclohexyl)-ureido]-butyric acid ethyl ester, and hesperetin dihydrochalcone – to the list of flavourings that can be used in foods.
EU authorises two flavourings: 3-[3-(2-isopropyl-5-methyl-cyclohexyl)-ureido]-butyric acid ethyl ester, and hesperetin dihydrochalcone
Commission Regulation (EU) 2026/172 of 26 January 2026 amending Annex I to Regulation (EC) No 1334/2008 of the European Parliament and of the Council as regards the inclusion of 3-[3-(2-isopropyl-5-methyl-cyclohexyl)-ureido]-butyric acid ethyl ester in the Union list of flavourings and correcting certain CAS numbers
Commission Regulation (EU) 2026/175 of 26 January 2026 amending Annex I to Regulation (EC) No 1334/2008 of the European Parliament and of the Council as regards the inclusion of hesperetin dihydrochalcone in the Union list of flavourings
Update
The European Union (EU) has added two substances – 3-[3-(2-isopropyl-5-methyl-cyclohexyl)-ureido]-butyric acid ethyl ester, and hesperetin dihydrochalcone – to the list of flavourings that can be used in foods.
Impacted Products
Flavoured fermented milk products (including heat-treated products), processed fruit and vegetables, confectionery, chewing gum, breakfast cereals, bread and rolls, herbs, spices, seasonings, sauces, alcoholic and non-alcoholic beverages (including alcohol-free and low-alcohol), potato-, cereal-, flour-, or starch-based snacks, cheese and cheese products, dairy analogues (including beverage whiteners), fats and oils, meat preparations, processed fish and fishery products, processed eggs and egg products, edible ices, beer and malt beverages
What is changing?
The EU has added the following to the list of auhorised flavourings that can be used in foods:
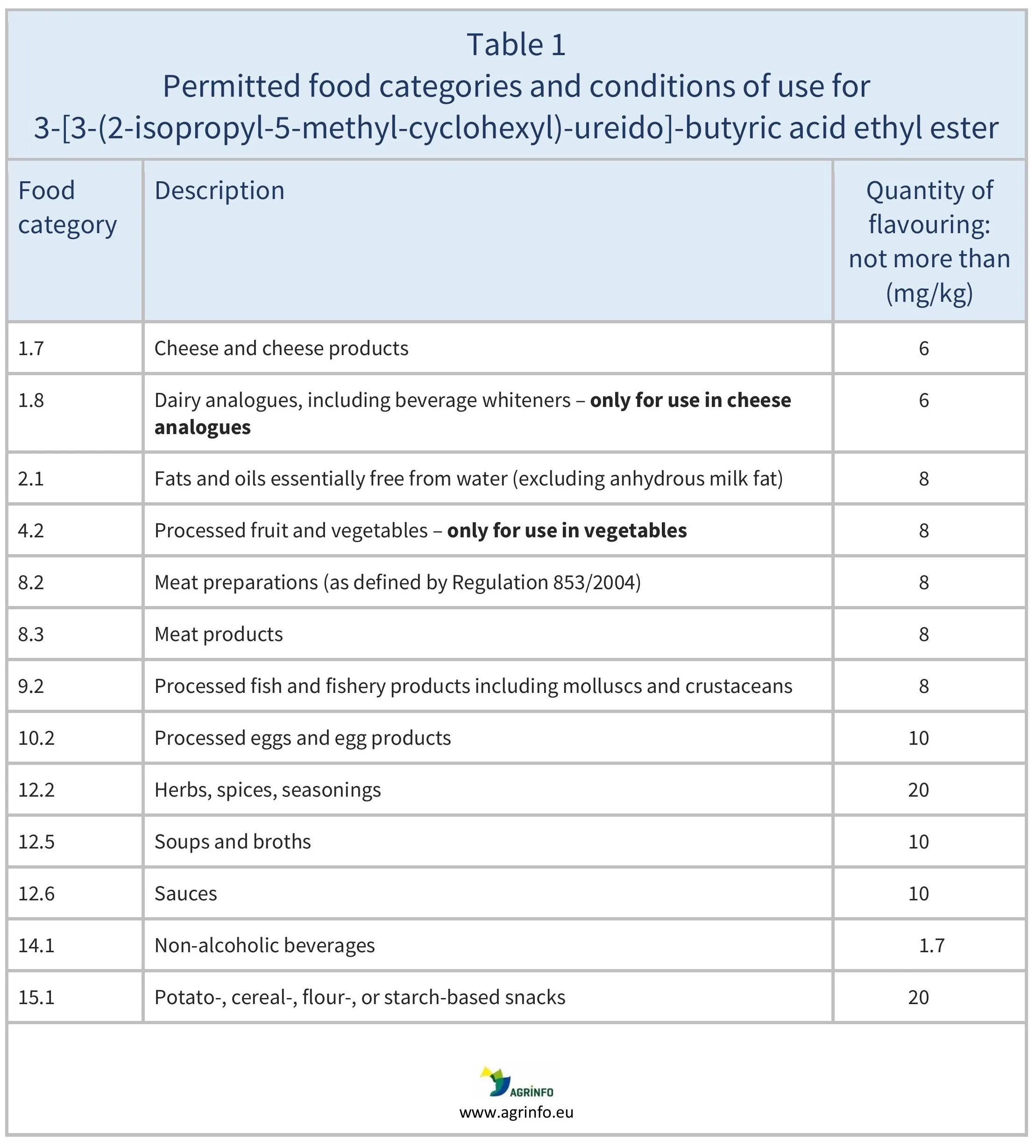
- [3-(2-isopropyl-5-methyl-cyclohexyl)-ureido]-butyric acid ethyl ester, for the uses given in Table 1
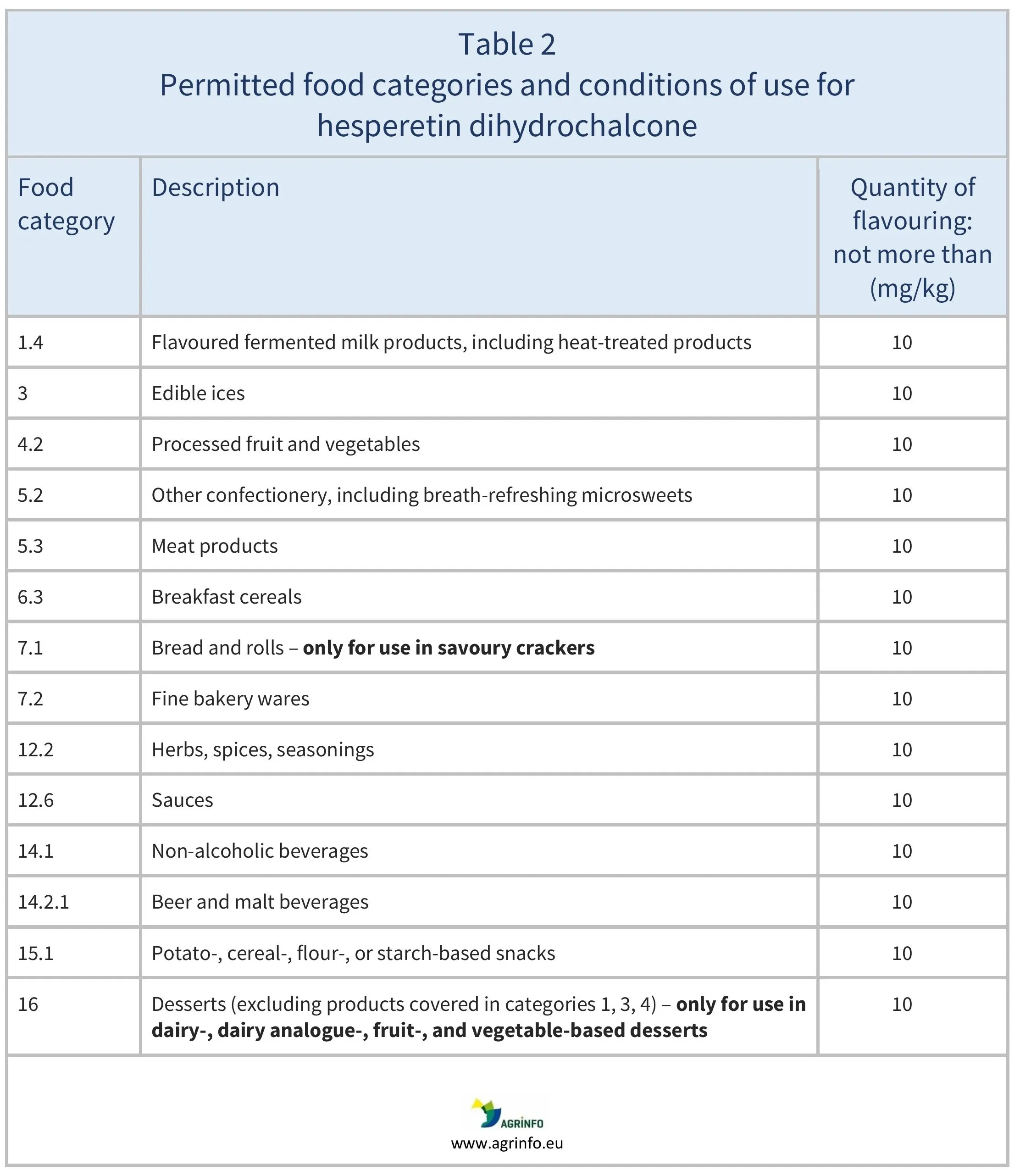
- hesperetin dihydrochalcone-one, for the uses given in Table 2.
For more information on the food categories used in these Tables, see guidance from the European Commission (2025).
Why?
The European Food Safety Authority (EFSA) evaluated the safety of 3-[3-(2-isopropyl-5-methyl-cyclohexyl)-ureido]-butyric acid ethyl ester and hesperetin dihydrochalcone under specific conditions in certain foods, and concluded that there is no safety concern at the levels proposed by the applicant (EFSA 2024, 2025).
Timeline
Products in Tables 1 and 2 containing 3-[3-(2-isopropyl-5-methyl-cyclohexyl)-ureido]-butyric acid ethyl ester and hesperetin dihydrochalcone, respectively, can be placed on the EU market from 15 February 2026.
Background
Regulation 1334/2008 prohibits adding certain undesirable natural substances to food. It also lays down maximum levels for some substances that are naturally present in flavourings, but that may raise concerns for human health. The Regulation defines different types of flavourings, and lists the substances for which evaluation and approval are required.
The Union list of flavouring substances approved for use in and on foods (Regulation 872/2012) was adopted in 2012.
When EFSA evaluates flavouring substances, it allocates a unique identification number called an FL-number. FL comes from “FLAVIS”, the EU’s flavouring information system. The FL numbers for these substances are:
- 3-[3-(2-isopropyl-5-methyl-cyclohexyl)-ureido]-butyric acid ethyl ester: 16.136
- hesperetin dihydrochalcone: 16.137.
The FL-number is not used for labelling purposes.
Resources
EFSA (2024) Flavouring group evaluation 420 (FGE.420): Hesperetin dihydrochalcone. EFSA Journal, 22(12): e9091.
EFSA (2025) Flavouring group evaluation 418 (FGE. 418): 3-[3-(2-isopropyl-5-methyl-cyclohexyl)-ureido]-butyric acid ethyl ester. EFSA Journal, 23(1): e9201.
European Commission (2025) Guidance document describing the food categories in Part E of Annex II to Regulation (EC) No 1333/2008 on Food Additives. Version 8.
Regulation 1331/2008 establishing a common authorisation procedure for food additives, food enzymes and food flavourings.
Regulation 1334/2008 on flavourings and certain food ingredients with flavouring properties for use in and on foods.
Sources
Commission Regulation (EU) 2026/172 as regards the inclusion of 3-[3-(2-isopropyl-5-methyl-cyclohexyl)-ureido]-butyric acid ethyl ester in the Union list of flavourings and correcting certain CAS numbers
Commission Regulation (EU) 2026/175 as regards the inclusion of hesperetin dihydrochalcone in the Union list of flavourings
Disclaimer: Under no circumstances shall COLEAD be liable for any loss, damage, liability or expense incurred or suffered that is claimed to have resulted from the use of information available on this website or any link to external sites. The use of the website is at the user’s sole risk and responsibility. This information platform was created and maintained with the financial support of the European Union. Its contents do not, however, reflect the views of the European Union.
EU authorises two flavourings: 3-[3-(2-isopropyl-5-methyl-cyclohexyl)-ureido]-butyric acid ethyl ester, and hesperetin dihydrochalcone
Commission Regulation (EU) 2026/172 as regards the inclusion of 3-[3-(2-isopropyl-5-methyl-cyclohexyl)-ureido]-butyric acid ethyl ester in the Union list of flavourings and correcting certain CAS numbers
Commission Regulation (EU) 2026/175 as regards the inclusion of hesperetin dihydrochalcone in the Union list of flavourings
What is changing and why?
The European Union (EU) has approved two flavourings – 3-[3-(2-isopropyl-5-methyl-cyclohexyl)-ureido]-butyric acid ethyl ester, and hesperetin dihydrochalcone – for use in certain types of food (see Tables 1 and 2).
Timeline
Products in Tables 1 and 2 containing 3-[3-(2-isopropyl-5-methyl-cyclohexyl)-ureido]-butyric acid ethyl ester, and hesperetin dihydrochalcone, respectively, can be placed on the EU market from 15 February 2026.
Disclaimer: Under no circumstances shall COLEAD be liable for any loss, damage, liability or expense incurred or suffered that is claimed to have resulted from the use of information available on this website or any link to external sites. The use of the website is at the user’s sole risk and responsibility. This information platform was created and maintained with the financial support of the European Union. Its contents do not, however, reflect the views of the European Union.