Latest pesticide approvals, renewals, and extensions 2024
- Food safety
- Pesticides
Summary
The most recent EU pesticide approval is for the basic substance Allium fistulosum. This report summarises new and renewed approvals so far in 2024. It also lists extensions of EU approval in 2024 (Table 1), and changes in conditions of use.
Latest EU pesticide approval: Allium fistulosum as basic substance
Commission Implementing Regulations 2014/2878; 2024/2198; 2024/2186; 2024/1892; 2024/1749; 2024/1718; 2024/1206; 2024/324; 2024/2781
Update
The most recent EU pesticide approval is for the basic substance Allium fistulosum. This report summarises new and renewed approvals so far in 2024. It also lists extensions of EU approval in 2024 (Table 1), and changes in conditions of use.
What is changing?
Approvals and renewals
The EU has recently approved the basic substance Allium fistulosum.
Previously in 2024, the EU renewed the approval of captan, folpet, and trinexapac; renewed the approval of metconazole as a candidate for substitution; and approved magnesium hydroxide E528 as a basic substance.
Extensions and amendments
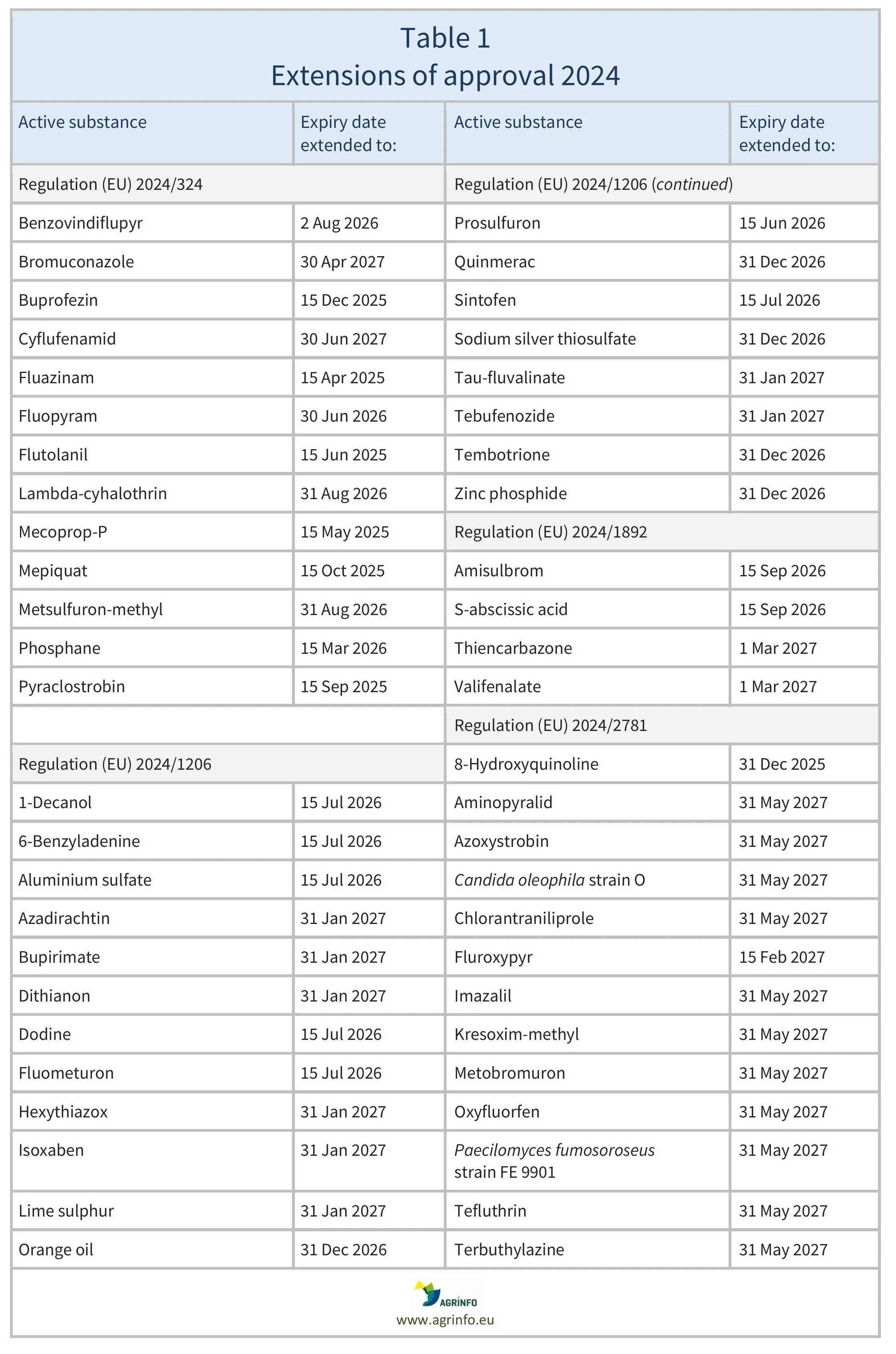
So far in 2024, the EU has extended approval of the substances listed in Table 1.
In addition, the EU has amended the conditions of approval of the active substance metalaxyl-M, which is now restricted to use as a treatment for seeds intended to be sown in greenhouses.
Timeline
A comprehensive list of active substances, their legal status, and expiry dates can be found in the European Commission’s Active substances, safeners and synergists database.
Recommended Actions
Suppliers to the EU market should monitor developments in EU approvals when considering longer-term strategies on agricultural practices that are in conformity with EU rules.
Background
Approvals
A plant protection product or pesticide contains one or several active substances. Active substances must meet the established approval criteria (Regulation 1107/2009, Art. 4), which include no harmful effects on human health or the environment.
Depending on their characteristics, some active substances can be approved as low-risk substances that fulfil certain criteria [Regulation 1107/2009, Annex II(5)]. Low-risk substances are approved for 15 years instead of the standard 10 years, and enjoy more favourable data protection (13 years). Authorisation procedures are generally also shorter.
Approval renewals
The Commission has established a series of work programmes to manage the renewal process. The current (fifth) work programme covers substances with approval expiring between 1 January 2022 and 31 December 2024. Each substance is allocated to an EU Member State for evaluation.
If no application is received for extension of approval, the initial expiry date will remain.
Extension of approvals
If an application for renewal cannot be fully evaluated before the expiry date, the approval can be extended to allow the evaluation to be completed.
Resources
Online resources from the European Commission:
Sources
Disclaimer: Under no circumstances shall COLEAD be liable for any loss, damage, liability or expense incurred or suffered that is claimed to have resulted from the use of information available on this website or any link to external sites. The use of the website is at the user’s sole risk and responsibility. This information platform was created and maintained with the financial support of the European Union. Its contents do not, however, reflect the views of the European Union.
Latest EU pesticide approval: Allium fistulosum as basic substance
Commission Implementing Regulations 2024/2878; 2024/2198; 2024/2186; 2024/1892; 2024/1749; 2024/1718; 2024/1206; 2024/324; 2024/2781
What is changing and why?
Approvals and renewals
The EU has recently renewed the approval of basic substance Allium fistulosum.
Previously in 2024, the EU renewed the approval of captan, folpet, and trinexapac; renewed the approval of metconazole as a candidate for substitution; and approved magnesium hydroxide E528 as a basic substance.
Extensions and amendments
So far in 2024, the EU has extended approval of the substances listed in Table 1.
In addition, the EU amended the conditions of approval of the active substance metalaxyl-M, which is now restricted to use as a treatment for seeds intended to be sown in greenhouses.
Actions
Suppliers to the EU market should monitor developments in EU approvals when developing longer-term strategies on agricultural practices that are in conformity with EU rules.
Timeline
A comprehensive list of active substances, their legal status, and expiry dates can be found on the European Commission’s Active substances, safeners and synergists database.
Disclaimer: Under no circumstances shall COLEAD be liable for any loss, damage, liability or expense incurred or suffered that is claimed to have resulted from the use of information available on this website or any link to external sites. The use of the website is at the user’s sole risk and responsibility. This information platform was created and maintained with the financial support of the European Union. Its contents do not, however, reflect the views of the European Union.