Maximum residue levels for thiacloprid
- Pesticide MRLs
- Pesticides
Summary
The EU has lowered the maximum residue levels (MRLs) for thiacloprid to the limit of determination (LOD) for all products. The LOD is the lowest level that can be detected using the most modern and reliable analytical methods.
In February 2024, France introduced a national law banning placement on the French market of fruit and vegetables containing thiacloprid at levels over the LOD of 0.01 mg/kg (République Française 2024), including products for which there is an EU MRL above the LOD. This national regulation states that it will be lifted with the adoption of any EU Regulation.
EU reduces MRLs for thiacloprid on many products
Commission Regulation (EU) 2024/2711 of 22 October 2024 amending Annexes II and V to Regulation (EC) No 396/2005 of the European Parliament and of the Council as regards maximum residue levels for thiacloprid in or on certain products
Update
The EU has lowered the maximum residue levels (MRLs) for thiacloprid to the limit of determination (LOD) for all products. The LOD is the lowest level that can be detected using the most modern and reliable analytical methods.
In February 2024, France introduced a national law banning placement on the French market of fruit and vegetables containing thiacloprid at levels over the LOD of 0.01 mg/kg (République Française 2024), including products for which there is an EU MRL above the LOD. This national regulation states that it will be lifted with the adoption of any EU Regulation.
Impacted Products
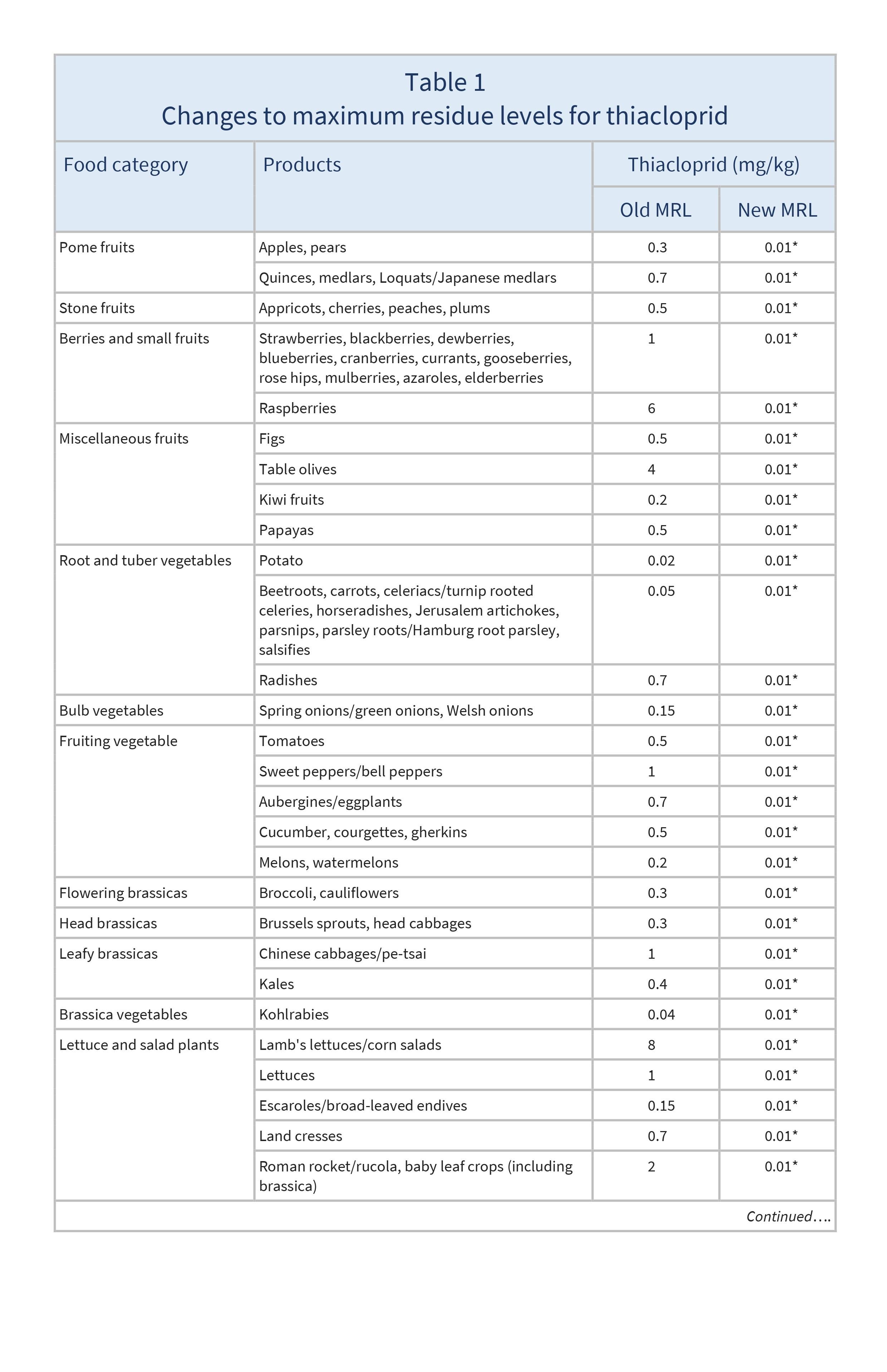
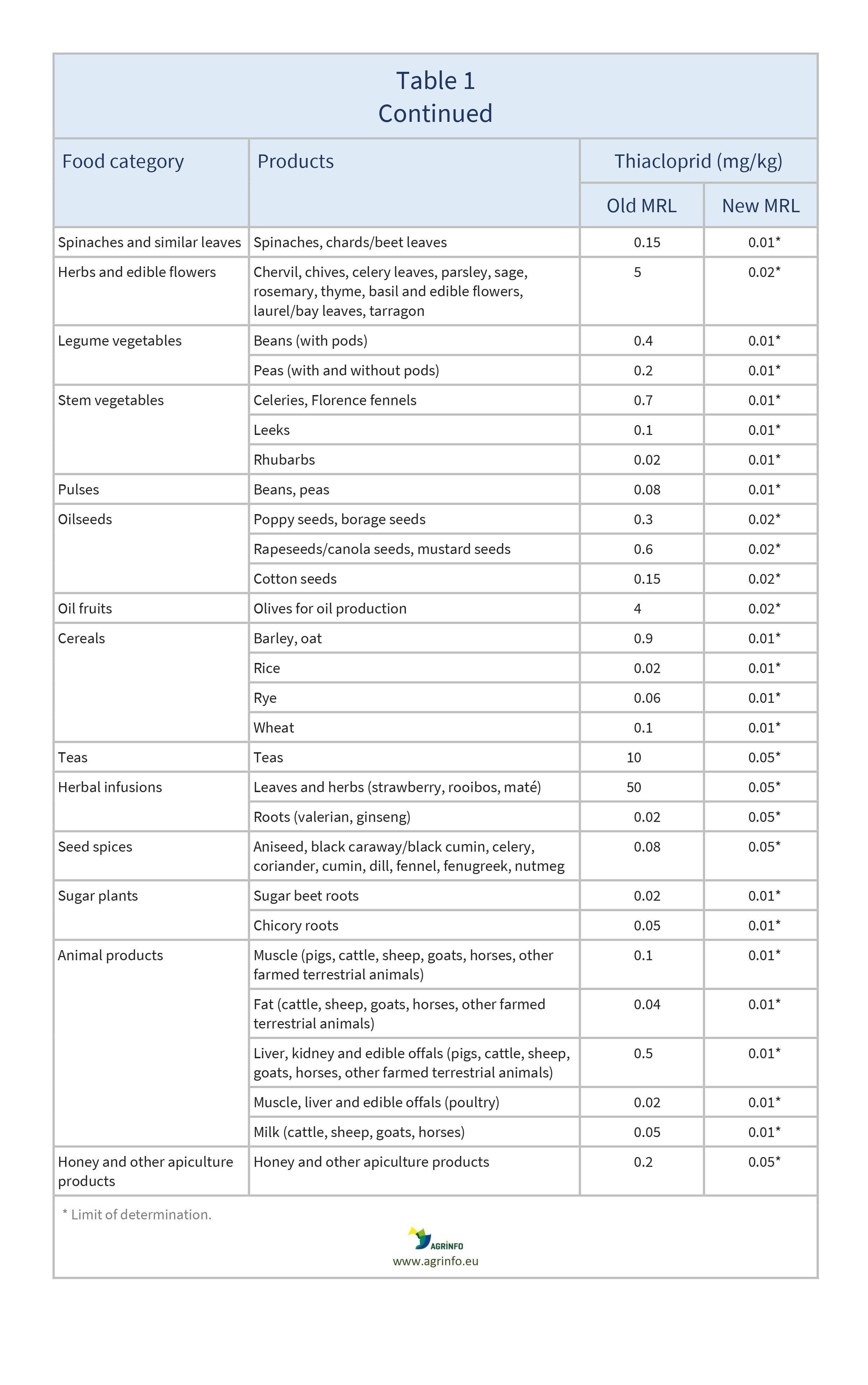
Apples, pears, quinces, medlars, loquats/ Japanese medlars, apricots, cherries, peaches, plums, strawberries, blackberries, dewberries, blueberries, cranberries, currants, gooseberries, rose hips, mulberries, azaroles, elderberries, raspberries, figs, table olives, kiwi fruits, papayas, potatoes, beetroots, carrots, celeriacs/ turnip rooted celeries, horseradishes, Jerusalem artichokes, parsnips, parsley roots/ Hamburg root parsley, salsifies, radishes, spring onions/ green onions, Welsh onions, tomatoes, sweet peppers/ bell peppers, aubergines/ eggplants, cucumbers, courgettes, gherkins, melons, watermelons, broccoli, cauliflowers, Brussels sprouts, head cabbages, Chinese cabbages/ pe-tsai, kales, kohlrabies, lamb's lettuces/ corn salads, lettuces, escaroles/ broad-leaved endives, land cresses, Roman rocket/ rucola, baby leaf crops (including brassicas), spinaches, chards/ beet leaves, chervil, chives, celery leaves, parsley, sage, rosemary, thyme, basil and edible flowers, laurel/ bay leaves, tarragon, beans (with pods), peas (with and without pods), celeries, Florence fennels, leeks, rhubarbs, beans, peas, poppy seeds, borage seeds, rapeseeds/ canola seeds, mustard seeds, cotton seeds, olives for oil production, barley, oat, rice, rye, wheat, teas, leaves and herbs (strawberry, rooibos, maté), roots (valerian, ginseng), aniseed, black caraway/ black cumin, celery, coriander, cumin, dill, fennel, fenugreek, nutmeg, sugar beet roots, chicory roots, muscle (pigs, cattle, sheep, goats, horses, other farmed terrestrial animals), fat (cattle, sheep, goats, horses, other farmed terrestrial animals), liver, kidney and edible offals (pigs, cattle, sheep, goats, horses, other farmed terrestrial animals), muscle, liver and edible offals (poultry), milk (cattle, sheep, goats, horses), honey and other apiculture products
What is changing?
The EU has reduced all MRLs for thiacloprid to the LOD (0.01–0.05 mg/kg).
The changes to EU MRLs for thiacloprid are shown in Table 1. All other products not mentioned in Table 1 were already set at the LOD.
Why?
The approval of thiacloprid for use in the EU was not renewed due to its toxicity and the risk of groundwater contamination. The Commission originally proposed that all MRLs for thiacloprid should be set to the LOD except where EFSA (2023) has concluded there are no health risks for consumers. In these cases (cucumber, courgette, rapeseeds, canola seeds, and mustard seeds), the Commission proposed maintaining import tolerances or Codex maximum residue levels (CXLs).
However, EFSA’s risk assessment was carried out before the most recent EU criteria on endocrine disruptors were adopted. The Commission considers there are reasonable grounds for concern about the potential endocrine-related effects of thiacloprid, and has therefore provisionally lowered the MRLs based on import tolerances until EFSA has completed an additional assessment of those effects.
Timeline
The new MRLs apply from 12 May 2025.
Recommended Actions
Exporters of all products should review their current use of thiacloprid and evaluate possible alternative solutions in anticipation of the MRL changes.
Background
MRLs are set in accordance with the rules set out in Regulation 396/2005. For information on current MRLs for other substances, please consult the EU Pesticide Residues database.
An initial proposed Regulation on thiacloprid MRLs which proposed maintaining an import tolerance MRL for cucumber, courgette, rapeseeds, canola seeds, and mustard seeds (G/SPS/N/EU/651) was rejected by the European Parliament (European Parliament 2024).
Resources
EFSA (2023) Statement on the short‐term (acute) dietary risk assessment and evaluation of confirmatory data for certain maximum residue levels (MRLs) for thiacloprid. EFSA Journal, 21(3): 7888.
European Parliament (2024) Objection to an implementing act: Maximum residue levels for thiacloprid.
Sources
Commission Regulation (EU) 2024/2711 as regards maximum residue levels for thiacloprid in or on certain products
Tables & Figures
Disclaimer: Under no circumstances shall COLEAD be liable for any loss, damage, liability or expense incurred or suffered that is claimed to have resulted from the use of information available on this website or any link to external sites. The use of the website is at the user’s sole risk and responsibility. This information platform was created and maintained with the financial support of the European Union. Its contents do not, however, reflect the views of the European Union.
EU reduces MRLs for thiacloprid on many products
Commission Regulation (EU) 2024/2711 as regards maximum residue levels for thiacloprid in or on certain products
What is changing and why?
The European Commission has reduced the maximum residue levels (MRLs) for thiacloprid to the limit of determination (LOD) for all products. The LOD is the lowest level that can be detected using the most modern and reliable analytical methods. These changes are due to concerns about risks to public health.
The changes to EU MRLs for thiacloprid are shown in Table 1.
In February 2024, France introduced a national law banning placement on the French market of fruit and vegetables containing thiacloprid at levels over the LOD of 0.01 mg/kg (République Française 2024). France indicated that it would lift the national regulation after the new EU Regulation is adopted.
Actions
Exporters of all products should review their current use of thiacloprid and evaluate possible alternative solutions in anticipation of the MRL changes.
Timeline
The new MRLs apply from 12 May 2025.
Tables & Figures
Disclaimer: Under no circumstances shall COLEAD be liable for any loss, damage, liability or expense incurred or suffered that is claimed to have resulted from the use of information available on this website or any link to external sites. The use of the website is at the user’s sole risk and responsibility. This information platform was created and maintained with the financial support of the European Union. Its contents do not, however, reflect the views of the European Union.