Maximum residue levels for napropamide
- Food safety
- Pesticide MRLs
Summary
The EU has decided to reduce the maximum residue levels (MRLs) for napropamide on some products to the limit of determination (LOD, the lowest level that can be detected using the most modern and reliable analytical methods). There will be potential impacts on exports of berries, herbs, and edible flowers.
EU reduces MRLs for napropamide, with impacts on berries, herbs, and edible flowers
Commission Regulation (EU) 2024/2609 of 7 October 2024 amending Annex II to Regulation (EC) No 396/2005 of the European Parliament and of the Council as regards maximum residue levels for napropamide, pyridaben and tebufenpyrad in or on certain products
Update
The EU has decided to reduce the maximum residue levels (MRLs) for napropamide on some products to the limit of determination (LOD, the lowest level that can be detected using the most modern and reliable analytical methods). There will be potential impacts on exports of berries, herbs, and edible flowers.
Impacted Products
Blueberries, cranberries, currants, gooseberries, rose hips, elderberries, chervil, chives, celery leaves, parsley, sage, rosemary, thyme, basil and edible flowers, laurel/bay leaves, tarragon
What is changing?
The EU has amended the MRLs for napropamide as summarised in Table 1.
Why?
The European Food Safety Authority (EFSA) reviewed the MRLs for napropamide and found gaps in the data needed to support safe MRLs for certain products. After the applicant provided additional information to address these gaps, most MRLs were maintained. However, EFSA (2023) recommended lowering the MRLs to the LOD on products where the additional information was not sufficient.
Timeline
The new MRLs apply from 28 April 2025.
Products exported before 28 April 2025 that comply with the old MRLs will not be removed from the EU market after April 2025, even if they do not comply with the new MRLs.
Recommended Actions
Exporters of certain berries, herbs, and edible flowers should review their current use of napropamide and evaluate possible alternative solutions in anticipation of MRL changes.
Background
MRLs are set in accordance with the rules set out in Regulation 396/2005. For information on current MRLs for other substances, please consult the EU Pesticide Residues database.
Resources
EFSA (2023) Evaluation of confirmatory data following the Article 12 MRL review for napropamide. EFSA Journal, 21(7): 8125.
Sources
Commission Regulation (EU) 2024/2609 as regards maximum residue levels for napropamide, pyridaben and tebufenpyrad in or on certain products
Tables & Figures
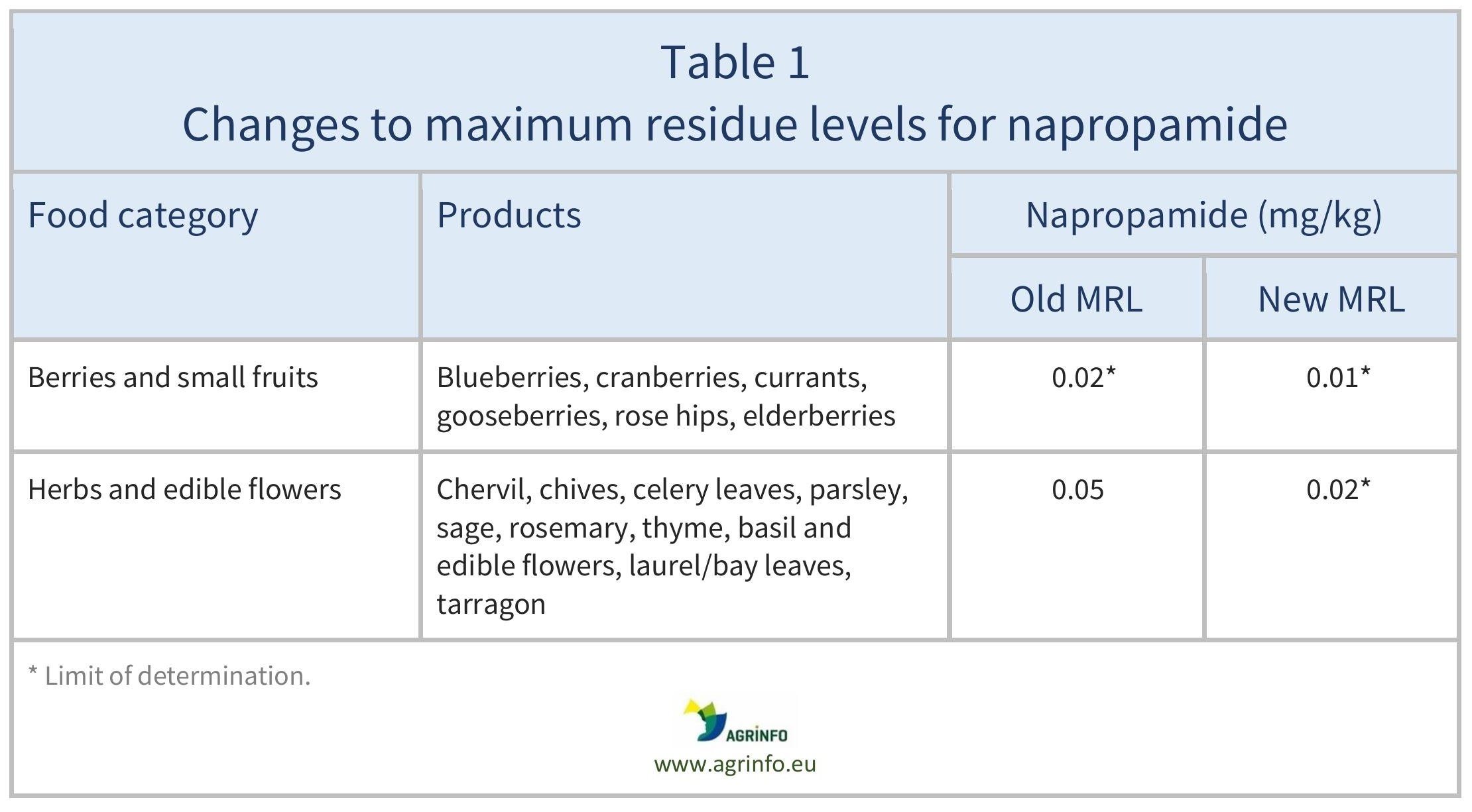
Source: based on Regulation (EU) 2024/2609
Disclaimer: Under no circumstances shall COLEAD be liable for any loss, damage, liability or expense incurred or suffered that is claimed to have resulted from the use of information available on this website or any link to external sites. The use of the website is at the user’s sole risk and responsibility. This information platform was created and maintained with the financial support of the European Union. Its contents do not, however, reflect the views of the European Union.
EU reduces MRLs for napropamide, with impacts on berries, herbs, and edible flowers
Commission Regulation (EU) 2024/2609 as regards maximum residue levels for napropamide, pyridaben and tebufenpyrad in or on certain products
What is changing and why?
The EU will lower the maximum residue levels (MRLs) for napropamide to the limit of determination (LOD) on some berries and small fruits, herbs, and edible flowers, as summarised in Table 1. (The LOD is the lowest level that can be detected using the most modern and reliable analytical methods.) This change is due to insufficient data to support the MRLs of those products.
Actions
Exporters of certain berries, herbs, and edible flowers should review their current use of napropamide and evaluate possible alternative solutions in anticipation of MRL changes.
Timeline
The new MRLs apply from 28 April 2025.
Products exported before 28 April 2025 that comply with the old MRLs will not be removed from the EU market after April 2025, even if they do not comply with the new MRLs.
Tables & Figures

Source: based on Regulation (EU) 2024/2609
Disclaimer: Under no circumstances shall COLEAD be liable for any loss, damage, liability or expense incurred or suffered that is claimed to have resulted from the use of information available on this website or any link to external sites. The use of the website is at the user’s sole risk and responsibility. This information platform was created and maintained with the financial support of the European Union. Its contents do not, however, reflect the views of the European Union.
