Maximum residue levels for thiophanate-methyl
- Food safety
- Pesticide MRLs
- Pesticides
Summary
The European Union (EU) has notified the World Trade Organization Sanitary and Phytosanitary Measures (WTO SPS) Committee of a new draft proposal to reduce the maximum residue levels (MRLs) for thiophanate-methyl to the limit of determination (LOD) for all products (G/SPS/N/EU/916). (The LOD is the lowest level that can be detected using the most modern and reliable analytical methods.)
This proposal follows the rejection by the European Parliament in September 2024 of a previous European Commission proposal to maintain MRLs on limes and okra. See Maximum residue levels for benomyl, carbendazim, thiophanate-methyl, cyproconazole, and spirodiclofen. The new proposal is a response to the Parliament’s request to the Commission to reduce the MRLs for thiophanate-methyl on all products to the LOD.
EU to reduce thiophanate-methyl MRLs to limit of determination on all products
Draft Commission Regulation amending Annex II, III and V to Regulation (EC) No 396/2005 of the European Parliament and of the Council as regards maximum residue levels for benomyl, carbendazim and thiophanate-methyl in or on certain products
Draft Annex V [PLAN/2024/2763 v6]
Update
The European Union (EU) has notified the World Trade Organization Sanitary and Phytosanitary Measures (WTO SPS) Committee of a new draft proposal to reduce the maximum residue levels (MRLs) for thiophanate-methyl to the limit of determination (LOD) for all products (G/SPS/N/EU/916). (The LOD is the lowest level that can be detected using the most modern and reliable analytical methods.)
This proposal follows the rejection by the European Parliament in September 2024 of a previous European Commission proposal to maintain MRLs on limes and okra. See Maximum residue levels for benomyl, carbendazim, thiophanate-methyl, cyproconazole, and spirodiclofen. The new proposal is a response to the Parliament’s request to the Commission to reduce the MRLs for thiophanate-methyl on all products to the LOD.
Impacted Products
All products
What is changing?
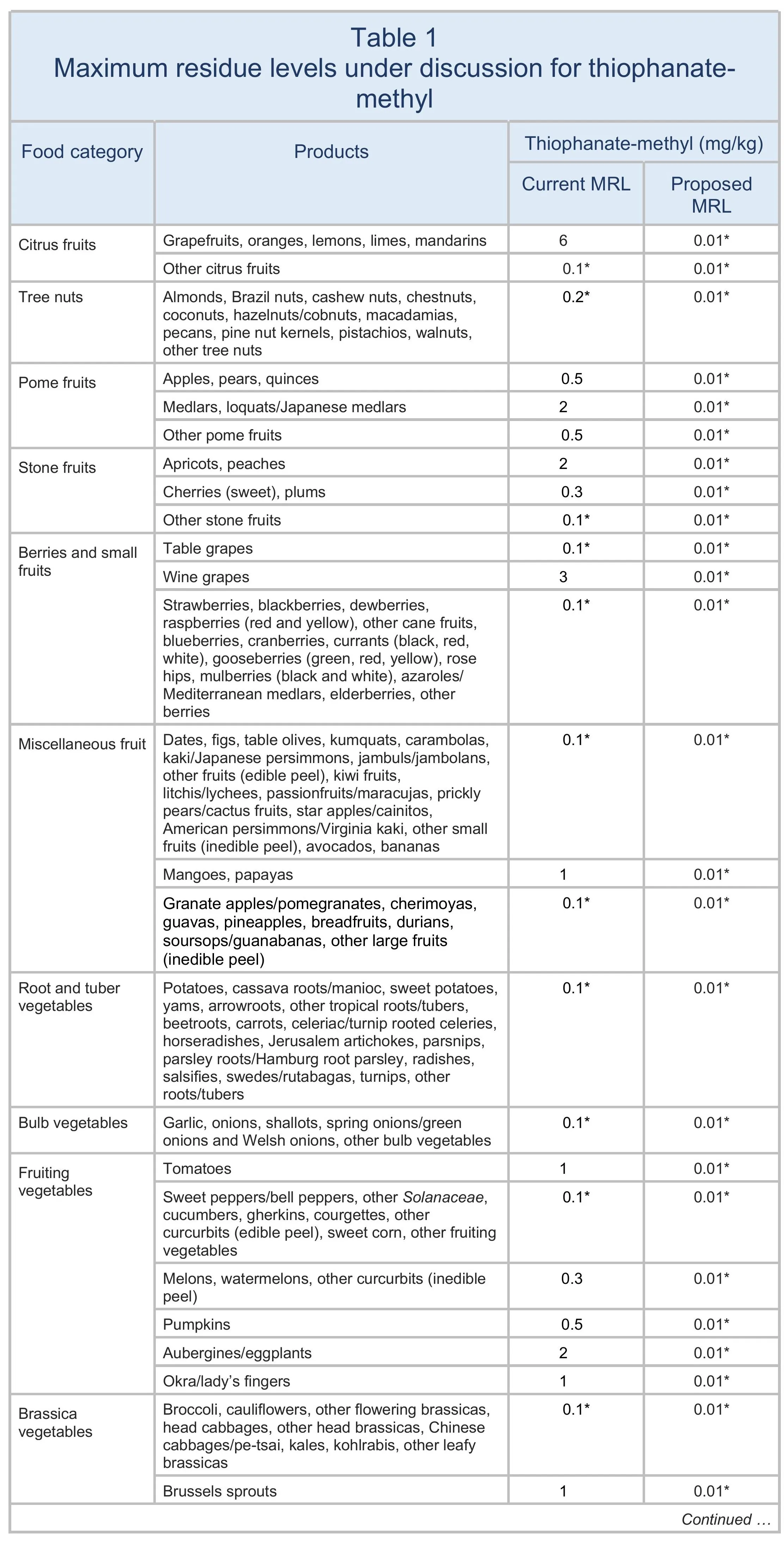
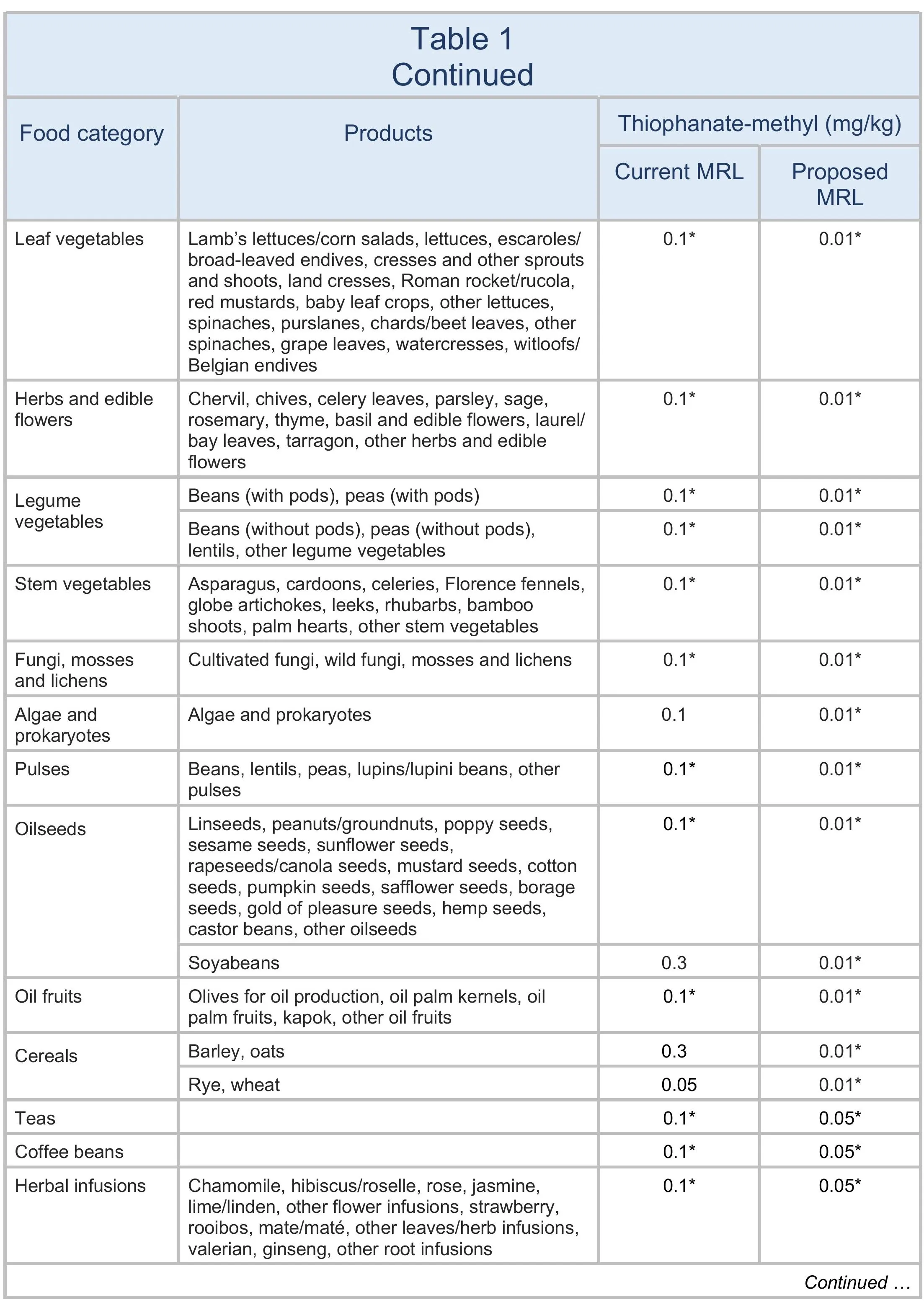
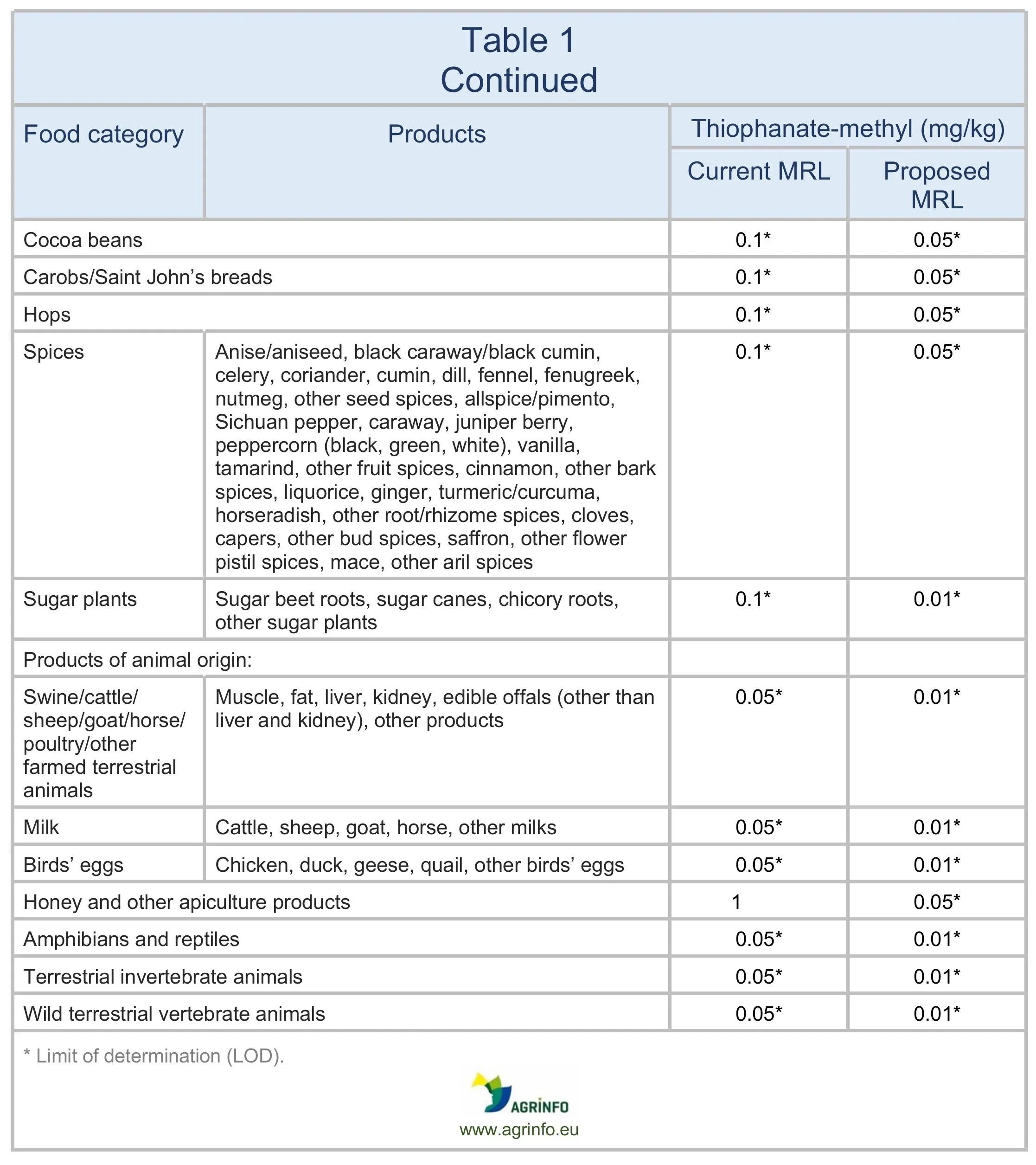
The EU proposes to lower the MRLs for thiophanate-methyl to 0.01 mg/kg on all products. The changes are summarised in Table 1.
For animal products, thiophanate-methyl is currently included in the definition of carbendazim: “carbendazim and thiophanate-methyl, expressed as carbendazim”. The proposal is to separate out thiophanate-methyl.
Why?
Thiophanate-methyl is no longer authorised in the EU because the manufacturer withdrew its new application for approval.
The European Food Safety Authority (EFSA 2021) suggested reducing the MRLs to the LOD, except on certain products for which it is considered safe. It proposed increasing the MRL for thiophanate-methyl on limes, aligning with good agricultural practices (GAPs) observed in certain non-EU countries (especially South Africa), and lowering the current MRL to a level deemed safe on okra/lady’s fingers, also derived from GAPs in non-EU countries.
However, the GAPs previously submitted for limes are no longer authorised in South Africa. In addition, there are concerns about the risks of combined residues from carbendazim and thiophanate-methyl. Regarding the lowering of the MRL for okra, there was a lack of information about “how these plant protection products are applied in practice” (recital [introductory paragraph] 10 of the proposed Regulation).
Timeline
The Regulation is expected to be published in July 2026 and will apply 6 months after publication.
Recommended Actions
Suppliers to the EU market of all products should seek alternative chemical and non-chemical alternatives to the use of thiophanate-methyl.
Competent authorities of countries that are members of the WTO can submit comments on the EU’s proposal by emailing the EU SPS Enquiry Point until 6 April 2026.
Background
In September 2024, the European Parliament rejected a European Commission Regulation that proposed to reduce the MRLs for thiophanate-methyl to the LOD on all products except limes and okra (see Maximum residue levels for benomyl, carbendazim, thiophanate-methyl, cyproconazole, and spirodiclofen). The Parliament requested the Commission to withdraw its draft Regulation and present a new one, setting the MRLs for thiophanate-methyl MRL on all products to the LOD.
MRLs are set in accordance with the rules set out in Regulation 396/2005. For information on current MRLs for other substances, please consult the EU Pesticide Residues database.
Resources
EFSA (2021) Reasoned opinion on the toxicological properties and maximum residue levels (MRLs) for the benzimidazole substances carbendazim and thiophanate-methyl. EFSA Journal, 19(8): e06773.
European Commission (2025a) Standing Committee on Plants, Animals, Food and Feed: Section Phytopharmaceuticals – Pesticide Residues, 17–18 February 2025. Summary Report.
European Commission (2025b) Standing Committee on Plants, Animals, Food and Feed: Section Phytopharmaceuticals – Pesticide Residues, 23–24 June 2025. Agenda.
Sources
Draft Commission Regulation amending Annex II, III and V to Regulation (EC) No 396/2005 of the European Parliament and of the Council as regards maximum residue levels for benomyl, carbendazim and thiophanate-methyl in or on certain products
Draft Annex V [PLAN/2024/2763 v6]
Tables & Figures
Disclaimer: Under no circumstances shall COLEAD be liable for any loss, damage, liability or expense incurred or suffered that is claimed to have resulted from the use of information available on this website or any link to external sites. The use of the website is at the user’s sole risk and responsibility. This information platform was created and maintained with the financial support of the European Union. Its contents do not, however, reflect the views of the European Union.
EU to reduce thiophanate-methyl MRLs to limit of determination on all products
Draft Commission Regulation amending Annex II, III and V to Regulation (EC) No 396/2005 of the European Parliament and of the Council as regards maximum residue levels for benomyl, carbendazim and thiophanate-methyl in or on certain products
Draft Annex V [PLAN/2024/2763 v6]
What is changing and why?
The European Union (EU) proposes to reduce the maximum residue levels (MRLs) for thiophanate-methyl to the limit of determination (LOD) for all products (see Table 1). (The LOD is the lowest level that can be detected using the most modern and reliable analytical methods.)
Actions
Suppliers to the EU market of all products should seek alternative chemical and non-chemical alternatives to the use of thiophanate-methyl.
Competent authorities of countries that are members of the World Trade Organization can submit comments on the EU’s proposal by emailing the EU SPS Enquiry Point until 6 April 2026.
Timeline
The Regulation is expected to be published in July 2026 and will apply 6 months after publication.
Tables & Figures
Disclaimer: Under no circumstances shall COLEAD be liable for any loss, damage, liability or expense incurred or suffered that is claimed to have resulted from the use of information available on this website or any link to external sites. The use of the website is at the user’s sole risk and responsibility. This information platform was created and maintained with the financial support of the European Union. Its contents do not, however, reflect the views of the European Union.