Official health certificates: food-producing horses and horse products
- Animal health
- Animal health certification
- Food safety
- Third country lists
- Veterinary residues
- Animal health controls
- Food safety controls
- Official controls
Summary
The European Commission has informed the World Trade Organization Sanitary and Phytosanitary Measures (WTO SPS) Committee (G/SPS/N/EU/891) that it intends to amend the official certificates required to export horses that will be used in food, and horse products, to the European Union (EU). The certificates need to be amended to accurately reflect the EU’s prohibition of the use of certain substances, such as thyrostatic (antithyroid) substances and stilbenes, which should apply not only to the exporting non-EU country, but also to the non-EU country where the horse was born and reared (if different).
EU to propose revised official health certificates for food-producing horses and horse products
Draft Commission Implementing Regulation amending Annex III to Implementing Regulation (EU) 2020/2235 and Annex II to Implementing Regulation (EU) 2021/403 as regards model certificates for entry into the Union of consignments of certain products of domestic solipeds origin intended for human consumption and certain categories of equine animals
Update
The European Commission has informed the World Trade Organization Sanitary and Phytosanitary Measures (WTO SPS) Committee (G/SPS/N/EU/891) that it intends to amend the official certificates required to export horses that will be used in food, and horse products, to the European Union (EU). The certificates need to be amended to accurately reflect the EU’s prohibition of the use of certain substances, such as thyrostatic (antithyroid) substances and stilbenes, which should apply not only to the exporting non-EU country, but also to the non-EU country where the horse was born and reared (if different).
Impacted Products
Horses for use in food, horse products
What is changing?
The EU is proposing changes to the official certificates required for the export to the European Union of horses that will be used in food, and horse products. This affects the following model certificates:
Details of the changes to these model certificates are given in Table 1.
Why?
The EU prohibits the use of certain substances, such as thyrostatic (antithyroid) substances and stilbenes, during the lifetime of a horse if the meat from that horse is intended to be consumed as food in the EU (Directive 96/22/EC). Non-EU countries involved in the production of horse meat must be listed in Regulation 2021/405, Annex -I (see Third country lists for public health – explained).
Currently the wording of the official certificates that must accompany consignments of these products reflects these requirements only for the exporting country. It does not cover a non-EU country where the horse was born and reared, if different to the exporting country. This draft Regulation intends to align the wording of the certificates to the requirements of Directive 96/22/EC, Art. 3 (a), and Art. 11.
Timeline
The amended certificates are expected to apply from 30 October 2026.
Recommended Actions
Where a non-EU country is exporting horses or their products that originated in another non-EU country, the official veterinarian signing the export certificate must check that this additional non-EU country fully complies with the EU rules (including non-use of prohibited substances), and is listed with an “X” for “equine” in Annex -I of Regulation 2021/405.
The WTO consultation closed on 30 December 2025.
Background
Council Directive 96/22/EC bans the use of the following substances in food-producing horses [Art. 3 (a)], and bans imports into the EU of horses (and their products) that have been administered these substances (Art. 11):
- Aristolochia spp. and preparations, chloramphenicol, chlorpromazine, colchicine, dapsone, dimetridazole, metronidazole, nitrofurans, ronidazole (substances listed in Regulation 37/2010, Annex, Table 2)
- thyrostatic substances, stilbenes, stilbene derivatives, their salts and esters, oestradiol 17β and its ester-like derivatives (Directive 96/22/EC, Annex II).
Other substances with action on hormones (oestrogenic, androgenic, or gestagenic action) and beta-agonists (used to enhance lean muscle gain) are also prohibited unless they are necessary for therapeutic treatments and/or animal husbandry (Directive 96/22/EC, Annex III).
Horse products can only be imported from countries listed with an “X” for “equine” in Annex -I of Regulation 2021/405, including the non-EU country where the horse was born and reared, if different from the exporting country. Countries that authorise the use of the above-mentioned substances in their legislation cannot be listed in this Annex (Directive 96/22/EC, Art. 11).
The rules establishing controls of residues of pharmacologically active substances were reviewed in 2022 (see Official controls of veterinary drug residues in products of animal origin and Official controls on the use of pharmacologically active substances and their residues).
For an overview of official certificates, see EU official health certificates for exports to the EU – explained.
Resources
Council Directive 96/22/EC concerning the prohibition on the use in stockfarming of certain substances having a hormonal or thyrostatic action and of beta-agonists
Commission Delegated Regulation (EU) 2022/1644 with specific requirements for the performance of official controls on the use of pharmacologically active substances authorised as veterinary medicinal products or as feed additives and of prohibited or unauthorised pharmacologically active substances and residues thereof
Commission Implementing Regulation (EU) 2022/1646 on uniform practical arrangements for the performance of official controls as regards the use of pharmacologically active substances authorised as veterinary medicinal products or as feed additives and of prohibited or unauthorised pharmacologically active substances and residues thereof, on specific content of multi-annual national control plans and specific arrangements for their preparation
European Commission (2025) Guidelines on EU requirements for entry of animals and products of animal origin – Control plans for residues of veterinary medicines, pesticides and contaminants
Sources
Draft Commission Implementing Regulation as regards model certificates for entry into the Union of consignments of certain products of domestic solipeds origin intended for human consumption and certain categories of equine animals
Tables & Figures
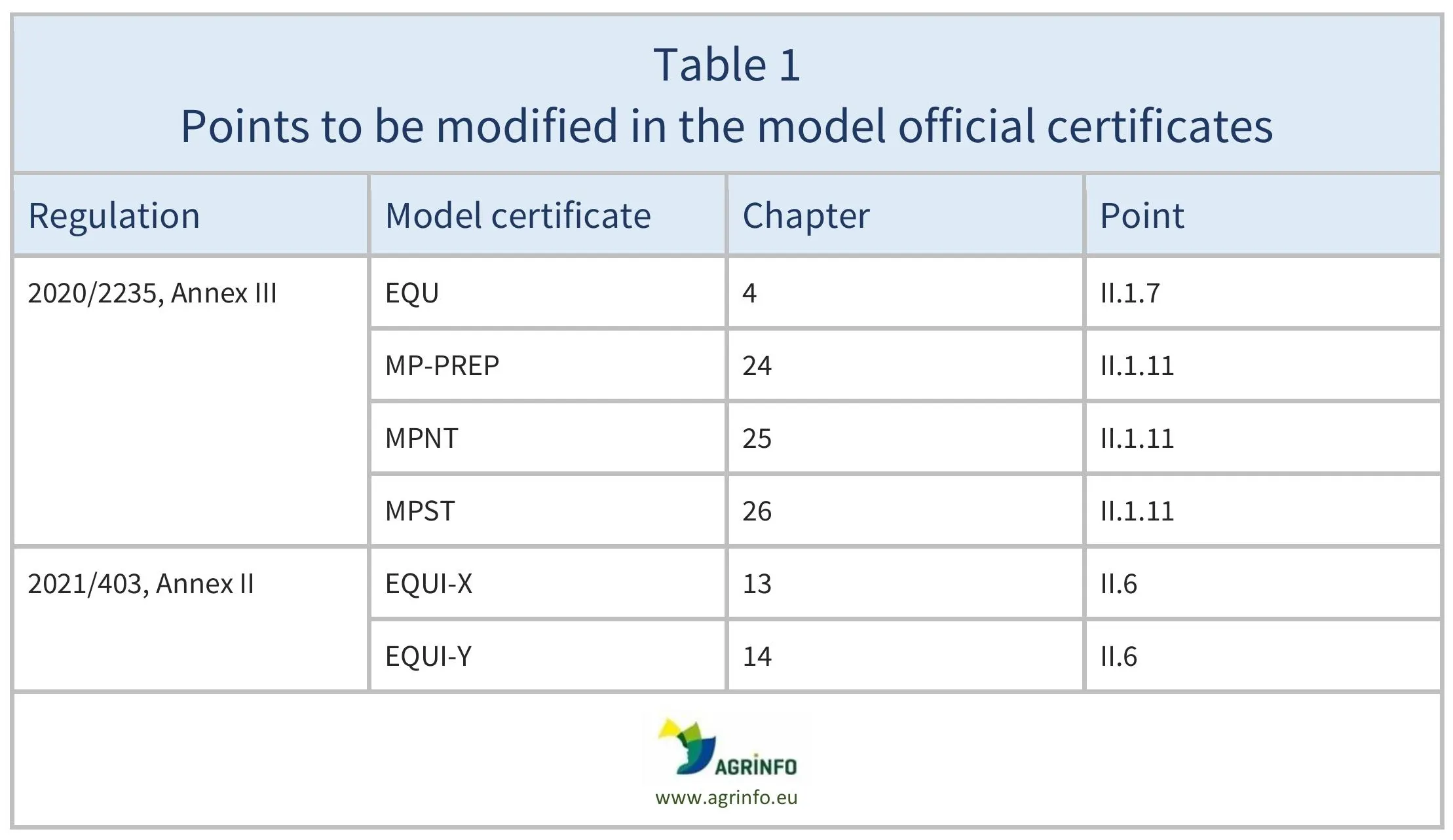
Source: Draft Regulation and Draft Annexes
Disclaimer: Under no circumstances shall COLEAD be liable for any loss, damage, liability or expense incurred or suffered that is claimed to have resulted from the use of information available on this website or any link to external sites. The use of the website is at the user’s sole risk and responsibility. This information platform was created and maintained with the financial support of the European Union. Its contents do not, however, reflect the views of the European Union.
EU to propose revised official health certificates for food-producing horses and horse products
Draft Commission Implementing Regulation as regards model certificates for entry into the Union of consignments of certain products of domestic solipeds origin intended for human consumption and certain categories of equine animals
What is changing and why?
The European Union (EU) prohibits the use of certain substances, such as thyrostatic (antithyroid) substances and stilbenes, during the lifetime of a horse if the meat from that horse is intended to be consumed as food in the EU. Non-EU countries involved in the production of horse meat must be listed in Regulation 2021/405, Annex-I.
The wording of the official certificates that must accompany consignments of these products currently reflects these requirements only for the exporting country; it does not include non-EU countries in which the horse was born and reared, if different from the exporting country. This draft Regulation intends to align the wording of the certificates so the requirements apply not only to the exporting country, but also to any non-EU country where the horse was born or reared (if different) - as described in Table 1.
Actions
Where a non-EU country is exporting horses or their products that originated in another non-EU country, the official veterinarian signing the export certificate must check that this additional non-EU country fully complies with the EU rules (including non-use of prohibited substances), and is listed with an “X” for “equine” in Annex-I of Regulation 2021/405.
The World Trade Organization (WTO) consultation closed on 30 December 2025.
Timeline
The amended certificates are expected to apply from 30 October 2026.
Tables & Figures

Source: Draft Regulation and Draft Annexes
Disclaimer: Under no circumstances shall COLEAD be liable for any loss, damage, liability or expense incurred or suffered that is claimed to have resulted from the use of information available on this website or any link to external sites. The use of the website is at the user’s sole risk and responsibility. This information platform was created and maintained with the financial support of the European Union. Its contents do not, however, reflect the views of the European Union.
