Maximum levels of delta-9-tetrahydrocannabinol (Δ9-THC) in hemp seeds
- Contaminants
Summary
The European Commission proposes to set a maximum level for delta-9-tetrahydrocannabinol (Δ9‐THC) in hemp leaves for water infusion and ready-to-drink herbal infusions made from hemp leaves to ensure a high level of human health protection.
Regulation 2023/915 sets maximum levels for (Δ9-THC) and delta-9-tetrahydrocannabinolic acid (Δ9-THCA) in hemp seeds and derived products sold on the European Union (EU) market, as severe exposure to Δ9‐THC might be a risk for human health.
EU proposes maximum levels for ∆9-THC in hemp leaves for infusion and ready-to-drink herbal infusions made from hemp leaves
Draft Commission Regulation (EU) amending Regulation (EU) 2023/915 as regards maximum levels of the sum of delta-9-tetrahydrocannabinol (Δ9-THC) in hemp leaves for water infusion and hemp leaves infusions
Draft Annex
Update
The European Commission proposes to set a maximum level for delta-9-tetrahydrocannabinol (Δ9‐THC) in hemp leaves for water infusion and ready-to-drink herbal infusions made from hemp leaves to ensure a high level of human health protection.
Regulation 2023/915 sets maximum levels for (Δ9-THC) and delta-9-tetrahydrocannabinolic acid (Δ9-THCA) in hemp seeds and derived products sold on the European Union (EU) market, as severe exposure to Δ9‐THC might be a risk for human health.
Impacted Products
Hemp seeds and derived products, hemp leaves for water infusion, ready-to-drink herbal infusions made from hemp leaves
What is changing?
The proposed maximum levels for the sum of Δ9-THC and Δ9-THCA, expressed as Δ9-THC, in hemp leaves for water infusion and ready-to-drink herbal infusions made from hemp leaves are presented in Table 1.
The European Commission proposes that hemp leaves for water infusion must be labelled with the following statements when sold in the EU:
- “Only to be used for water infusion (preparation of a herbal infusion).
- Shall not be consumed by infants and young children.
- No fatty ingredients, such as cream or milk, shall be added during steeping.”
In addition to these proposed maximum levels for Δ9-THC, EU countries can set more restrictive rules on their markets for selling hemp leaves and ready-to-drink infusions made from hemp leaves.
Why?
In 2015, the European Food Safety Authority (EFSA) assessed the risk to human health related to the presence of ∆9-THC in milk and dairy products (EFSA 2015).
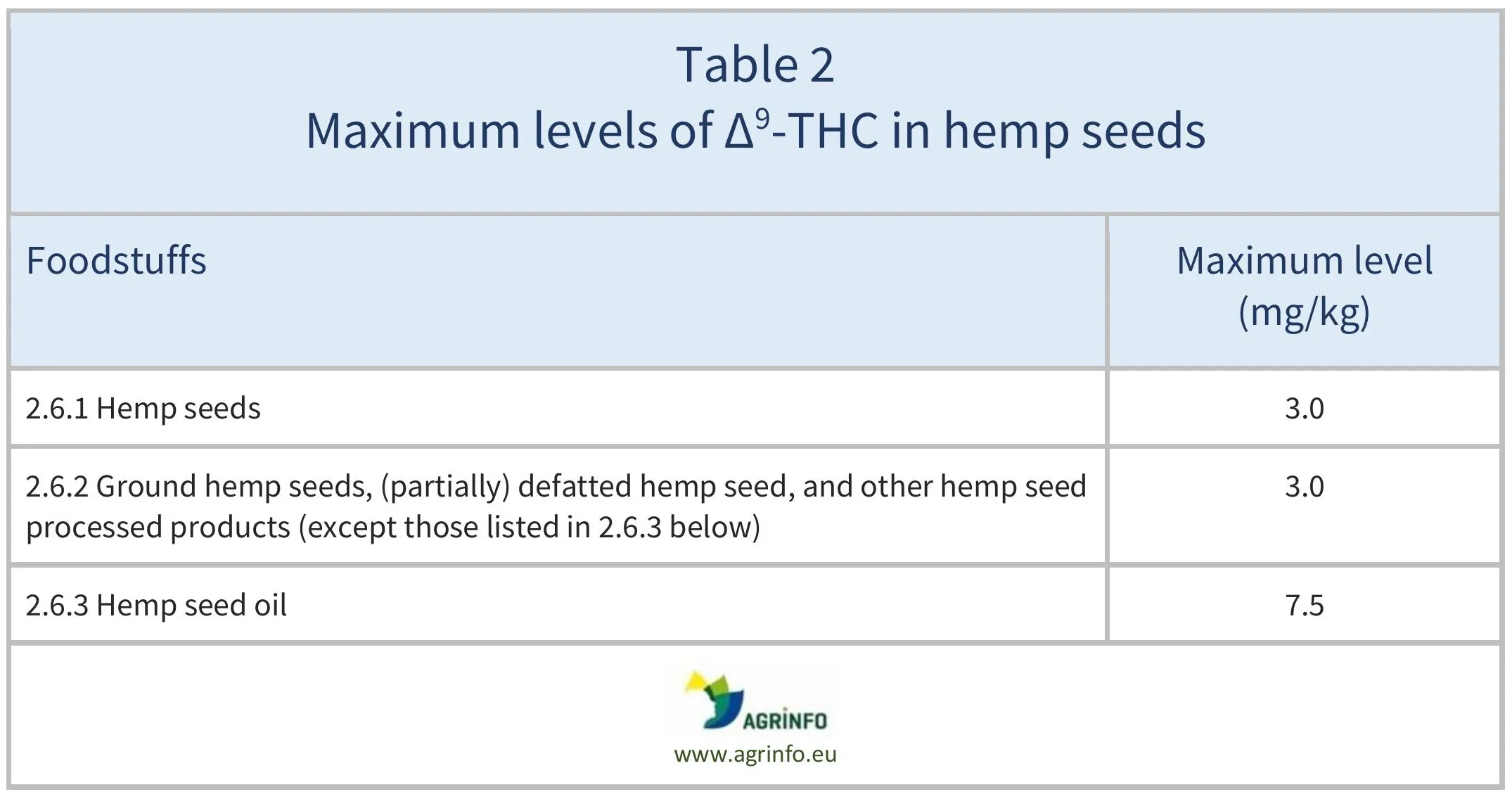
EFSA (2020) published a scientific report assessing exposure to ∆9-THC, and found that the reference dose of 1 μg ∆9-THC per kg body weight was exceeded in certain exposure estimates. The European Commission therefore considered that maximum levels should be set for hemp seeds and derived products (see Regulation 2023/915 and Table 2).
The European Commission now proposes to establish maximum levels for Δ9‐THC in hemp leaves for water infusion and ready-to-drink herbal infusions to ensure a high level of human health protection. Currently, water infusions of hemp leaves (when not accompanied by the flowering and fruiting tops), consumed as such or as part of herbal infusions, can be sold in the EU without prior authorisation, which might expose consumers to high levels of Δ9‐THC.
Timeline
This proposal is expected to be adopted in July 2026.
The proposed maximum levels for ∆9-THC in hemp leaves for water infusion and ready-to-drink herbal infusions are expected to apply from 1 January 2027.
Foodstuffs lawfully placed on the market before this Regulation enters into force may remain on the market until their date of minimum durability or use-by date.
Recommended Actions
Exporters of hemp seeds and derived products must ensure that adequate measures are put in place to prevent contamination during processing in order to meet the newly established levels (BfR 2018).
Competent authorities of countries that are members of the World Trade Organization can submit comments on the EU’s proposal by emailing the EU SPS Enquiry Point until 26 May 2026.
Resources
BfR (2018) Tetrahydrocannabinol levels are too high in many hemp-containing foods - health impairments are possible. Bundesinstitut für Risi.
Commission Regulation (EU) 2023/915 on maximum levels for certain contaminants in food
EFSA (2015) Scientific Opinion on the risks for human health related to the presence of tetrahydrocannabinol (THC) in milk and other food of animal origin. EFSA Journal, 13(6): 4141.
EFSA (2020) Acute human exposure assessment to tetrahydrocannabinol (Δ9‐THC). EFSA Journal, 18(1): 5953.
Sources
Disclaimer: Under no circumstances shall COLEAD be liable for any loss, damage, liability or expense incurred or suffered that is claimed to have resulted from the use of information available on this website or any link to external sites. The use of the website is at the user’s sole risk and responsibility. This information platform was created and maintained with the financial support of the European Union. Its contents do not, however, reflect the views of the European Union.
EU proposes maximum levels for ∆9-THC in hemp leaves for infusion and ready-to-drink herbal infusions made from hemp leaves
Draft Commission Regulation as regards maximum levels of the sum of delta-9-tetrahydrocannabinol (Δ9-THC) in hemp leaves for water infusion and hemp leaves infusions
Draft Annex
What is changing and why?
The European Commission proposes to set maximum levels for the sum of delta-9-tetrahydrocannabinol (Δ9-THC) and delta-9-tetrahydrocannabinolic acid (Δ9-THCA), expressed as Δ9-THC, in hemp leaves for water infusion and ready-to-drink herbal infusions made from hemp leaves (see Table 1). The aim is to ensure a high level of human health protection when adults consume these products.
Regulation 2023/915 sets the maximum levels for hemp seeds and derived products (see Table 2).
Actions
Exporters of hemp seeds and derived products must ensure that adequate measures are put in place to prevent contamination during processing in order to meet the newly established levels.
Competent authorities of countries that are members of the World Trade Organization can submit comments on the EU’s proposal by emailing the EU SPS Enquiry Point until 26 May 2026.
Timeline
The proposed maximum levels for Δ9‐THC in hemp leaves for water infusion and ready-to-drink herbal infusions are expected to apply from 1 January 2027.
Foodstuffs lawfully placed on the market before this Regulation enters into force may remain on the market until their date of minimum durability or use-by date.
Disclaimer: Under no circumstances shall COLEAD be liable for any loss, damage, liability or expense incurred or suffered that is claimed to have resulted from the use of information available on this website or any link to external sites. The use of the website is at the user’s sole risk and responsibility. This information platform was created and maintained with the financial support of the European Union. Its contents do not, however, reflect the views of the European Union.