Temporary increased official controls on food of certain origins
Published by AGRINFO on ; Revised
EU increases official controls on certain foods from specific non-EU countries
Commission Implementing Regulation (EU) 2024/286 of 16 January 2024 amending Implementing Regulation (EU) 2019/1793 on the temporary increase of official controls and emergency measures governing the entry into the Union of certain goods from certain third countries implementing Regulations (EU) 2017/625 and (EC) No 178/2002 of the European Parliament and of the Council
Update
The EU introduces Regulations to temporarily increase official controls on certain food products from specific countries when a potential risk to public health is identified. The latest such Regulation brings in increased controls for products from some countries, as well as reducing controls for others where the risk has reduced. The countries for which official controls have changed are: Bangladesh, Egypt, India, Madagascar, Pakistan, Sri Lanka, Thailand, Türkiye, Uganda, and Vietnam. An updated list of all increased official controls is provided in the Annex to Regulation 2024/286.
Impacted Products
black-eyed beans, capsicum peppers, cumin seeds, durian, granadilla, guar gum, helmet bean, passion fruit, rice, sesamum seeds, seem bean, vine leaves, yardlong beans
What is changing?
Implementing Regulation (EU) 2019/1793 lays down the list of food and feed of non-animal origin subject to a temporary increase of official controls, and the rules for these controls.
This list is updated regularly according to the risk of contamination by mycotoxins (including aflatoxins), pesticide residues, pentachlorophenol and dioxins, microbiological contamination, Sudan dyes, Rhodamine B, and plant toxins.
The details of the temporary increases are given in:
- Annex I of the Regulation, which lists products from certain non-EU countries that are subject to a temporary increase of official controls at EU border control posts and control points
- Annex II, which lists products and their countries of origin that are subject to special conditions as well as to a temporary increase of official controls on entering the EU.
The listing in the Annexes is very specific: each item refers to a particular product (e.g. groundnut) for an identified hazard (e.g. mycotoxins) from a named country.
Being listed in Annex I or Annex II has significant implications for suppliers. Annex II listing requires laboratory analysis and checks by competent authorities in exporting countries for each consignment of the affected commodities (see below).
Changes to Annex II
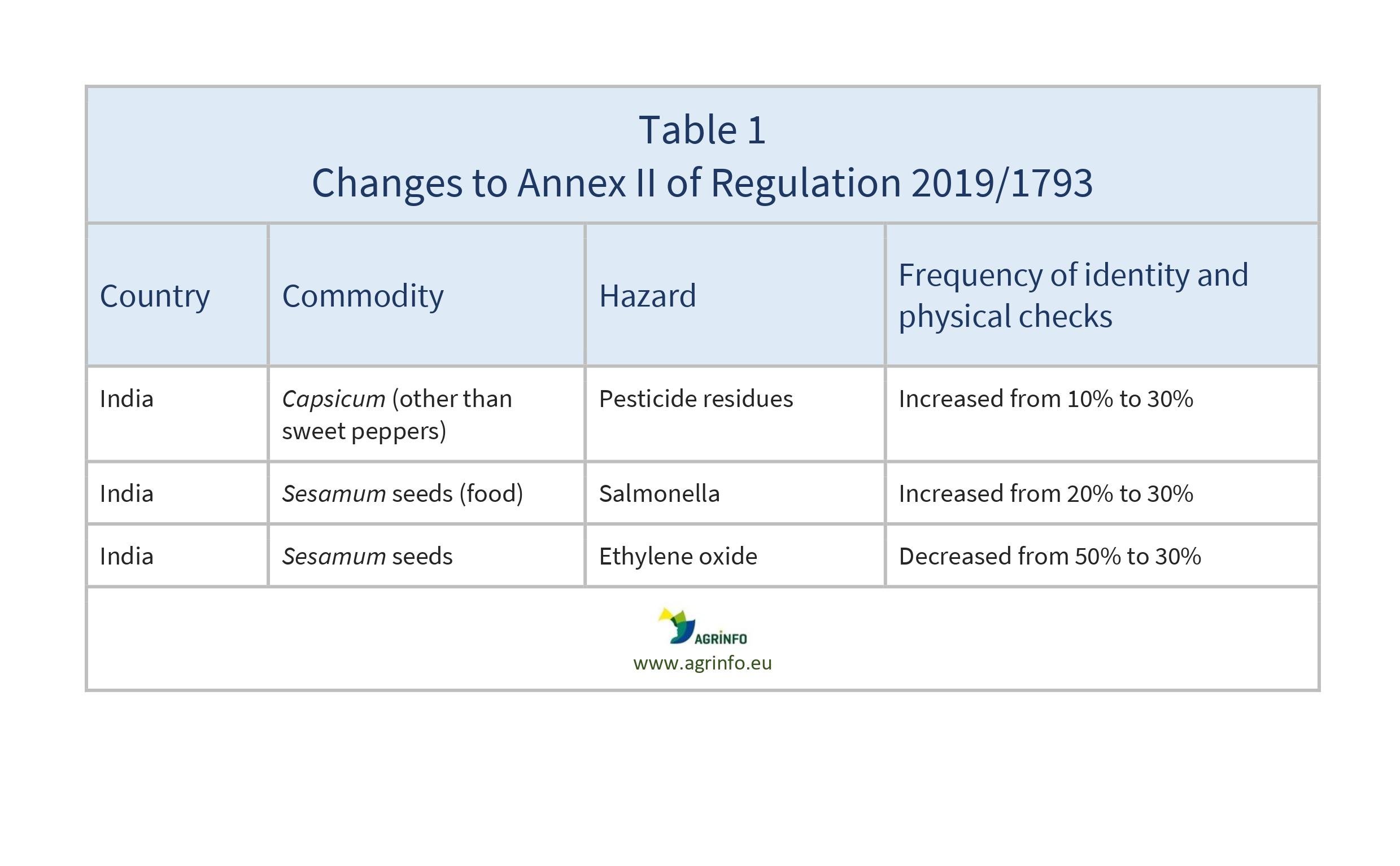
This new update of Regulation 2019/1793 makes a number of changes under Annex II affecting India (see Table 1 for details).
The following products from India have increased frequency of identity checks (visual inspection to ensure documentation corresponds with the products) and physical checks (checks on the product including sampling and laboratory testing):
- peppers of the genus Capsicum (other than sweet peppers) for pesticide residues
- Sesamum seeds for Salmonella.
The following from India have decreased frequency of identity and physical checks:
- Sesamum seeds for ethylene oxide.
Changes to Annex I
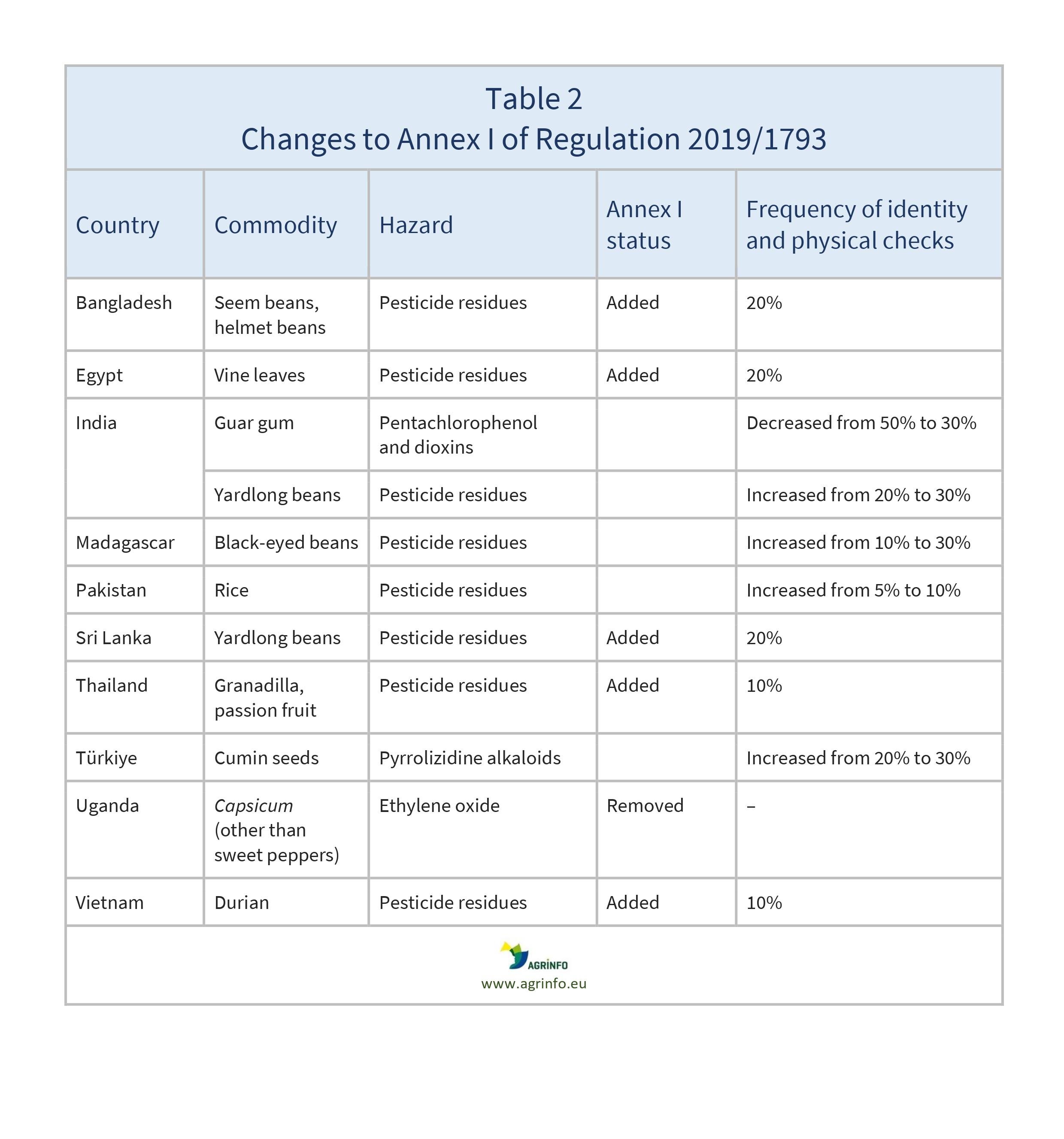
This new update of Regulation 2019/1793 makes the following changes to the list of consignments of food and feed subject to a temporary increase of official controls at EU border control posts (see Table 2 for details).
The following have been added to the list in Annex I:
- seem beans, helmet beans from Bangladesh for pesticide residues
- vine leaves from Egypt for pesticide residues
- yardlong beans from Sri Lanka for pesticide residues
- granadilla and passion fruit from Thailand for pesticide residues
- durian from Vietnam for pesticide residues.
The following have increased frequency of identity and physical checks:
- yardlong beans from India for pesticide residues
- black-eyed beans from Madagascar for pesticide residues
- rice from Pakistan for pesticide residues
- cumin seeds from Türkiye for pyrrolizidine alkaloids.
The following have decreased frequency of identity and physical checks:
- guar gum from India for pentachlorophenol and dioxins.
The EU has lifted temporary increased controls on the following products:
- Capsicum (other than sweet peppers) from Uganda for ethylene oxide.
For specific information on the products concerned (e.g. relevant customs codes, details of hazards that need to be analysed), and a complete list of all products/ product origins subject to increased frequency of controls, see the Annex of Regulation (EU) 2024/286.
Why?
The EU assesses the potential risks, and the associated need for additional controls, based on information from various sources. These include: data from EU Member State authorities and exporting countries; Commission audits carried out in exporting countries; and notifications reported to the Rapid Alert System for Food and Feed (RASFF) and Trade Control and Expert System (TRACES-NT). This information is normally reviewed every 6 months.
For commodities that are already listed, in consultation with Member States, the European Commission decides on a case-by-case basis whether risks have changed as a result of action taken by exporting countries, and if the increased controls or the special conditions can be relaxed or removed entirely. Further information on this decision-making process is provided in the Commission Notice on information related to risks and non-compliance.
Timeline
The changes to official controls apply from 5 February 2024.
What are the major implications for exporting countries?
Temporary increases of official controls place significant additional demands on private sector operators and competent authorities in exporting countries.
Implications of being put in Annex I
Increased controls create greater uncertainty in supply because of delays and possible rejections of consignments. This may weaken the reputation and competitive position of suppliers from a listed country. It also may have cost implications when operators must cover all or part of the cost of increased controls (e.g. storage, inspection, analysis, or destruction of consignments in an EU Member State).
Operators and competent authorities in countries supplying products listed in Annex I (additional controls) must ensure that they put in place the necessary measures to prevent any new non-compliances, and thus avoid a further increase in controls or listing in Annex II.
What does it mean to be put in Annex II instead of Annex I?
Annex II listing is triggered when there is evidence of a serious risk to human health, leading to stricter entry conditions for affected food and feed products exported to the EU.
The following actions must be taken in the country of origin (or the country from which the product is dispatched) before export for consignments of Annex II-listed food and feed.
- Each consignment must be accompanied by the results of sampling and laboratory analyses conducted by the competent authorities in the country of origin, or the country where it is dispatched from, if different.
- Laboratory analyses must be performed by laboratories that are accredited with the ISO/IEC 17025 Standard.
- Each consignment must have an identification code, and each individual bag or packaging in the consignment must be identified with that code.
- Each consignment must be accompanied by an official certificate issued by the competent authority of the exporting country, or the non-EU country where it is dispatched from, if different. This certificate must be based on a specific model and must state the identification code, must be issued before the consignment leaves the control of the competent authority, and must be valid for not more than 4 months from the date of issue, and no longer than 6 months from the date of the results of laboratory analyses.
- Suppliers of products listed in Annex II may face practical problems and high costs when they try to access appropriate testing facilities, particularly if there is no in-country accredited laboratory. Competent authorities have to put in place all the necessary procedures before listed consignments can be exported. They may also face technical and administrative constraints that make it difficult to respond to requests for mandatory certification. In practice, the listing of products in Annex II can lead to a temporary stop in exports from the countries concerned.
- Suppliers must ensure rigorous routine analysis of contaminants and pesticides to avoid listing in either of these Annexes.
Recommended Actions
Exporters of all products listed in Annexes I and II of Regulation (EU) 2024/286 must take steps to ensure full compliance with EU law.
Exporters of the following products newly added to Annex I should urgently review current practices and adopt additional measures to ensure compliance:
- seem beans and helmet beans from Bangladesh for pesticide residues
- vine leaves from Egypt for pesticide residues
- yardlong beans from Sri Lanka for pesticide residues
- granadilla and passion fruit from Thailand for pesticide residues
- durian from Vietnam for pesticide residues.
Resources
Online resources from the European Commission:
- Rapid Alert System for Food and Feed (RASFF)
- Trade Control and Expert System (TRACES-NT)
European Commission (2022) Notice on information related to risks and non-compliance in the context of periodic reviews of Commission Implementing Regulation (EU) 2019/1793.
Sources
Commission Implementing Regulation (EU) 2024/286 on the temporary increase of official controls and emergency measures governing the entry into the Union of certain goods from certain third countries
Implementing Regulation 2019/1793
Regulation (EU) 2017/625
Regulation (EC) No 178/2002
Tables & Figures