Managing non-authorised substances under the EU Organic Regulation
- Organic production
Summary
The European Union (EU) Organic Regulation 2018/848 places a strong emphasis on ensuring the integrity of organic and in-conversion agri-food products throughout the supply chain. Article 29 of the Organic Regulation concerns the measures to be taken in the EU when residues of non-authorised substances are detected, including in products imported from non-EU countries. This new report from the European Commission presents information from EU Member States in 2022–2024 about their official investigations into contamination of organic and in-conversion products. It also summarises findings of a dedicated study by the European Food Safety Authority (EFSA) on the 21 non-authorised substances most frequently detected in 2021–2022. Overall, the Commission concludes that the EU organic control system is robust and functioning effectively in this respect, and no major regulatory changes are considered necessary at this stage. The focus is on improving harmonisation, sharing best practices, and strengthening cooperation among stakeholders to maintain confidence in the EU organic label.
European Commission publishes report on the presence and management of non-authorised substances in organic products (2022–2024)
Report from the Commission to the European Parliament and the Council on the implementation of Article 29 of Regulation (EU) 2018/848 of the European Parliament and of the Council on organic production and labelling of organic products, on the presence of products and substances not authorised pursuant to the first subparagraph of Article 9(3) of said Regulation (EU) 2018/848 for use in organic production and on the assessment of the national rules referred to in paragraph 5 of Article 29
Commission Staff Working Document Accompanying the document Report from the Commission to the European Parliament and the Council on the implementation of Article 29 of Regulation (EU) 2018/848
Update
The European Union (EU) Organic Regulation 2018/848 places a strong emphasis on ensuring the integrity of organic and in-conversion agri-food products throughout the supply chain. Article 29 of the Organic Regulation concerns the measures to be taken in the EU when residues of non-authorised substances are detected, including in products imported from non-EU countries. This new report from the European Commission presents information from EU Member States in 2022–2024 about their official investigations into contamination of organic and in-conversion products. It also summarises findings of a dedicated study by the European Food Safety Authority (EFSA) on the 21 non-authorised substances most frequently detected in 2021–2022. Overall, the Commission concludes that the EU organic control system is robust and functioning effectively in this respect, and no major regulatory changes are considered necessary at this stage. The focus is on improving harmonisation, sharing best practices, and strengthening cooperation among stakeholders to maintain confidence in the EU organic label.
Impacted Products
Organic products
What is changing?
The Organic Regulation (EU) 2018/848 governs organic production and labelling, with a strong emphasis on ensuring the integrity of organic and in-conversion products throughout the supply chain. In the context of integrity of organic products, the presence of residues of non-authorised substances at any stage of the supply chain must be investigated. Article 29 of the Organic Regulation concerns the measures to be taken in the EU when non-authorised substances are detected, including in products imported from non-EU countries.
This new report from the European Commission builds on information submitted by EU Member States about their official investigations into contamination of organic and in-conversion products between 2022 and 2024. It also presents the findings of a study by EFSA on the 21 non-authorised substances notified in the Organic Farming Information System (OFIS) as being detected most frequently in organic products during 2021 and 2022 (EFSA 2025). The EFSA study confirms that organic products are significantly less likely than conventional products to contain pesticide residues. In 2023, 80% of organic samples showed no quantifiable residues, and only 0.9% exceeded the maximum residue levels (MRLs). However, even low-level detections require investigation to determine their origin and to exclude intentional use or insufficient preventive measures.
EFSA assessed the main potential sources of contamination, including soil residues, irrigation water, spray drift from conventional farming, and biocides. The main sources of contamination were identified as spray drift from conventional farming (24.1%), lack of precautionary measures by operators (16.7%), use of non-authorised substances (15.9%), and contamination at earlier stages of the supply chain (10.6%) (Table 1). Table 2 indicates the stage of the supply chain when the substances were detected.
Overall, the report concludes that the EU organic control system is robust and functioning effectively. While operational challenges remain, particularly regarding investigation timelines, costs, and consistency across authorities, no major regulatory changes are thought to be necessary at this stage. The focus will be on improving harmonisation, sharing best practices, and strengthening cooperation among stakeholders to ensure continued confidence in the EU organic label.
Why?
According to the Organic Regulation (Art. 29(4)), the European Commission must present a report to the European Parliament and the Council of the EU assessing the rules in place in individual EU Member States, and the measures taken when non-authorised substances are found to be present in organic and in-conversion products.
Timeline
The European Commission’s report was published on 31 March 2026.
What are the major implications for exporting countries?
During an official investigation, the products concerned are normally prohibited from being placed on the market as organic or in-conversion pending the results of the investigation. This can be problematic in the case of fresh products with a short shelf life.
The duration of official investigations varies significantly. According to this European Commission report, in more than half of EU Member States the average duration is from 20 to 30 days, while in others it ranges from 40 to 90 days. Reasons for requiring more time to complete official investigations include the need for additional sampling; incomplete reports; the complexity of the supply chain (type and number of operators involved); and the need for cooperation between different authorities.
Where contamination is linked to non-compliance (e.g. use of prohibited substances or inadequate preventive measures), the product cannot be marketed as organic. Operators are required to implement corrective actions, and failure to do so may lead to suspension or withdrawal of certification.
Recommended Actions
It is critical to avoid any risk of contamination in organic and in-conversion products for export to the EU. This report highlights best practices, including strict separation of organic and non-organic products, use of buffer zones, proper cleaning of equipment, risk-based sampling, and continuous training of operators and control bodies. Strong traceability and internal controls are essential to prevent and manage contamination risks.
Background
In the EU, the production and labelling of organic products are governed by the Organic Regulation (2018/848), in application since January 2022, and by detailed rules set out in numerous items of secondary legislation. Rules concerning the active substances authorised for use in organic and in-conversion products are set out in Annex I to Regulation 2021/1165. Organic products are also subject to the provisions of Regulations 396/2005 and 2017/625 that govern official controls and residues of pesticides in food and feed.
Article 29 of the Organic Regulation sets out the measures to be taken in the event that the presence of non-authorised substances is identified. To ensure the integrity of organic products, detection of non-authorised substances at any stage of the supply chain must trigger a mandatory official investigation. This is regardless of the residue level, and independent of food safety thresholds such as MRLs, reflecting the compliance-based (rather than threshold-based) approach.
Investigations aim to identify both the source and the cause of contamination. During this process, products are typically blocked from being marketed as organic. The duration of investigations varies, commonly between 20 and 30 days, depending on factors such as supply chain complexity and the need for additional sampling.
Resources
Commission Implementing Regulation (EU) 2021/1165 authorising certain products and substances for use in organic production and establishing their lists
EFSA (2025) Findings of not authorised substances in food and feed certified as organic. EFSA Supporting Publications, 22(7): EN-9524.
Regulation (EU) 2018/848 on organic production and labelling of organic products
Regulation (EU) 2017/625 on official controls and other official activities performed to ensure the application of food and feed law, rules on animal health and welfare, plant health and plant protection products
Regulation (EC) No 396/2005 on maximum residue levels of pesticides in or on food and feed of plant and animal origin
Sources
Report from the Commission to the European Parliament and the Council on the implementation of Article 29 of Regulation (EU) 2018/848 on organic production and labelling of organic products, on the presence of products and substances not authorised […] for use in organic production and on the assessment of the national rules
Commission Staff Working Document Accompanying the document Report from the Commission to the European Parliament and the Council on the implementation of Article 29 of Regulation (EU) 2018/848
Tables & Figures
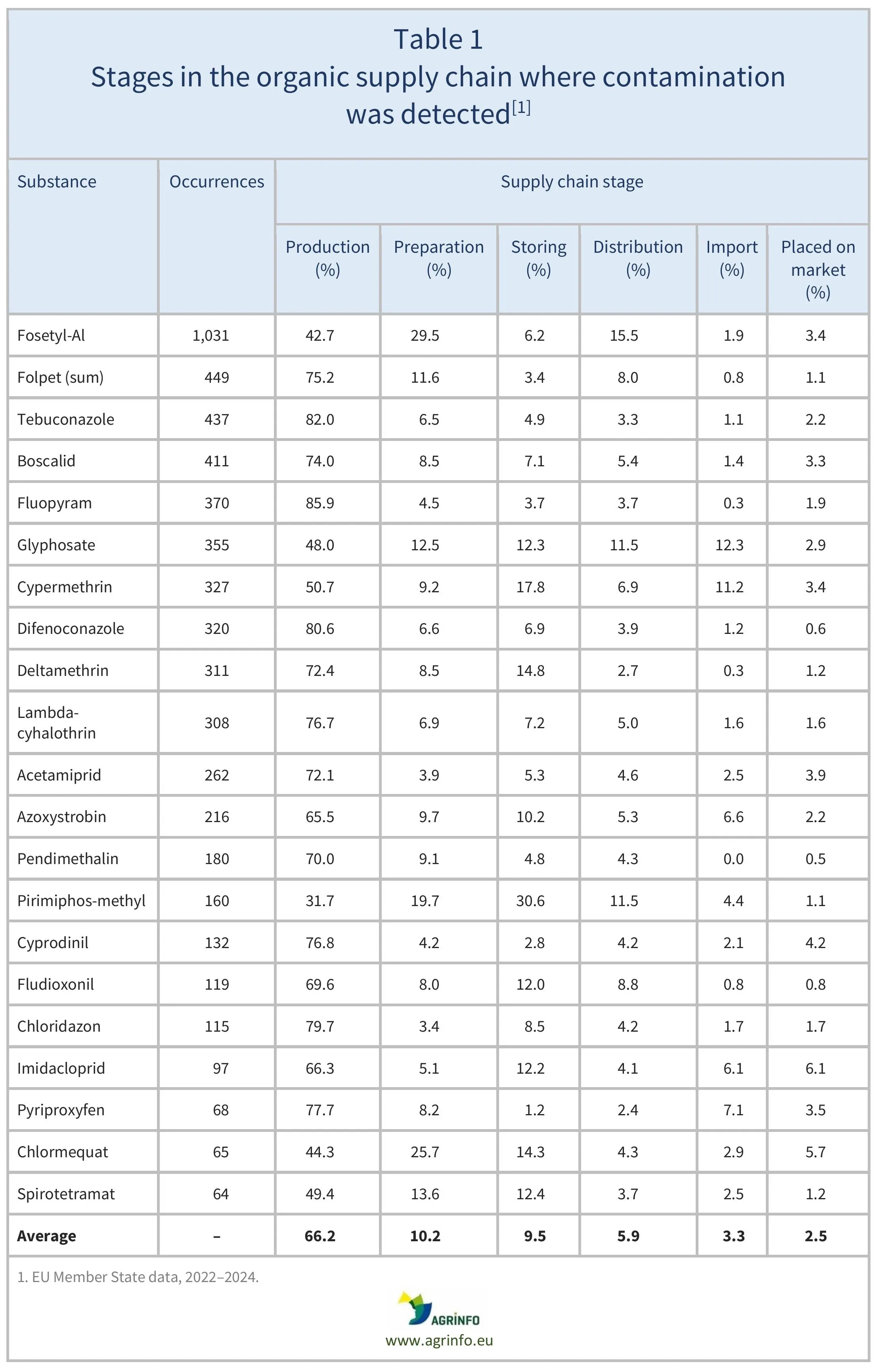
Source: based on Commission Staff Working Document Accompanying the Report on the implementation of Art. 29 of the Organic Regulation
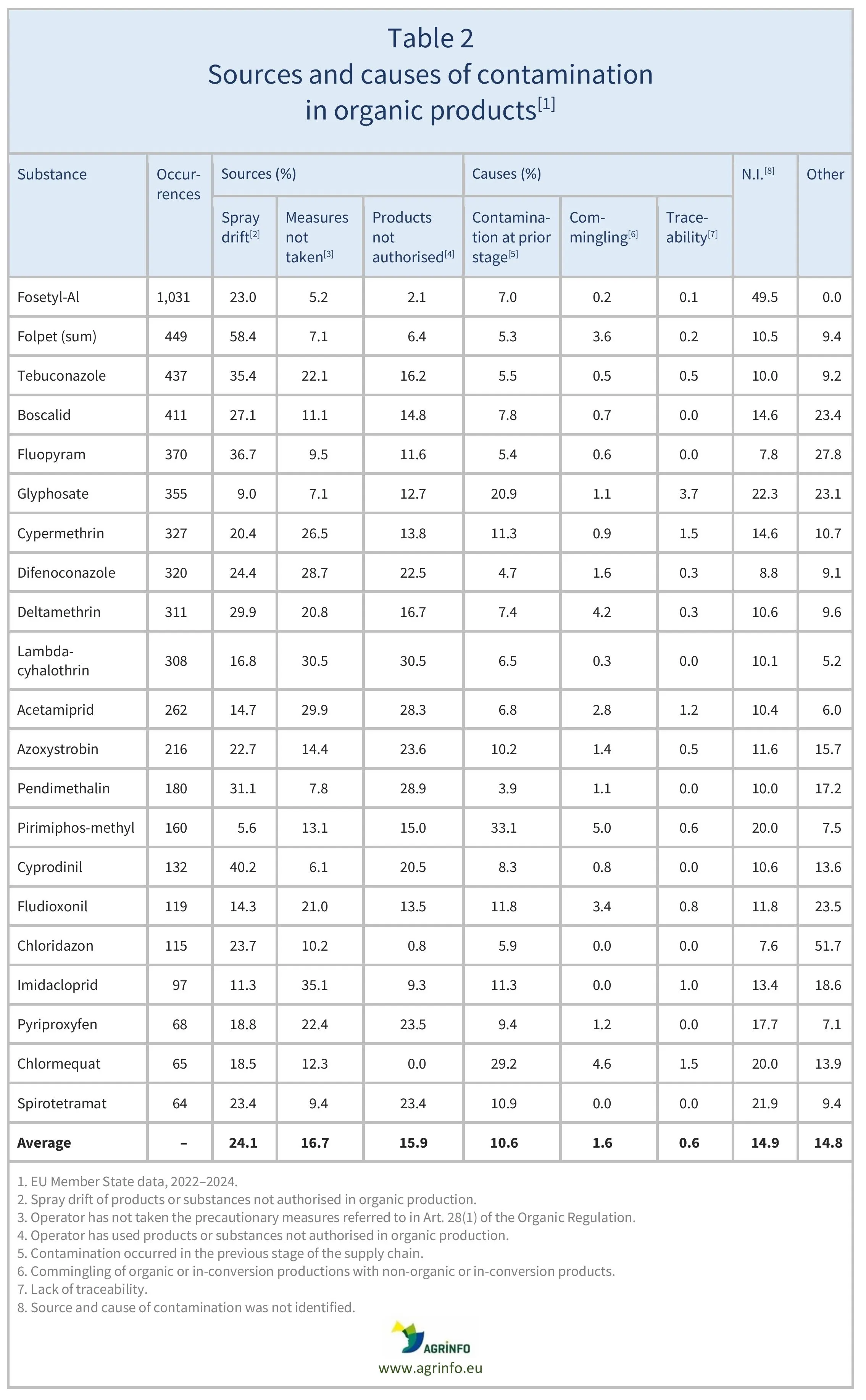
Source: based on Commission Staff Working Document Accompanying the Report on the implementation of Art. 29 of the Organic Regulation
Disclaimer: Under no circumstances shall COLEAD be liable for any loss, damage, liability or expense incurred or suffered that is claimed to have resulted from the use of information available on this website or any link to external sites. The use of the website is at the user’s sole risk and responsibility. This information platform was created and maintained with the financial support of the European Union. Its contents do not, however, reflect the views of the European Union.
European Commission publishes report on the presence and management of non-authorised substances in organic products (2022–2024)
Report from the Commission to the European Parliament and the Council on the implementation of Article 29 of Regulation (EU) 2018/848 on organic production and labelling of organic products, on the presence of products and substances not authorised […] for use in organic production and on the assessment of the national rules
Commission Staff Working Document Accompanying the document Report from the Commission to the European Parliament and the Council on the implementation of Article 29 of Regulation (EU) 2018/848
What is changing and why?
The European Union (EU) Organic Regulation 2018/848 governs organic production and labelling, with a strong emphasis on ensuring the integrity of organic products throughout the supply chain. In the context of integrity of organic products, the presence of residues of non-authorised substances at any stage of the supply chain must be investigated. This new report by the European Commission addresses the measures taken in the EU when non-authorised substances are detected, including in products imported from non-EU countries. The report presents information collected from individual EU countries, and from a study by the European Food Safety Authority (EFSA) about non-authorised substances detected in organic labelled products.
EFSA investigated the 21 non-authorised substances most frequently detected in organic products to better understand the sources of contamination. These included spray drift from conventional farming (24.1%), lack of precautionary measures by operators (16.7%), use of non-authorised substances (15.9%), and contamination at earlier stages of the supply chain (10.6%) (Table 1). Table 2 indicates the stage of the supply chain when the substances were detected.
The EFSA study confirms that organic products are significantly less likely than conventional products to contain pesticide residues. Overall, the European Commission concludes that the EU organic control system is robust and functioning effectively. While operational challenges remain, particularly regarding investigation timelines, costs, and consistency across authorities, no major regulatory changes are thought to be necessary at this stage. The focus will be on improving harmonisation, sharing best practices, and strengthening cooperation among stakeholders to ensure continued confidence in the EU organic label.
Actions
During an official investigation into the presence of contaminants, the products affected cannot normally be placed on the EU market as organic or in-conversion pending the results of the investigation. This can be problematic in the case of fresh products with a short shelf life. Where contamination is linked to non-compliance (e.g. use of prohibited substances or inadequate preventive measures), the product cannot be marketed as organic. Operators are required to implement corrective actions, and failure to do so may lead to suspension or withdrawal of certification.
It is critical to avoid any risk of contamination in organic and in-conversion products for export to the EU. This report highlights best practices, including strict separation of organic and non-organic products, use of buffer zones, proper cleaning of equipment, risk-based sampling, and continuous training of operators and control bodies. Strong traceability and internal controls are essential to prevent and manage contamination risks.
Timeline
The European Commission’s report was published on 31 March 2026.
Tables & Figures

Source: based on Commission Staff Working Document Accompanying the Report on the implementation of Art. 29 of the Organic Regulation

Source: based on Commission Staff Working Document Accompanying the Report on the implementation of Art. 29 of the Organic Regulation
Disclaimer: Under no circumstances shall COLEAD be liable for any loss, damage, liability or expense incurred or suffered that is claimed to have resulted from the use of information available on this website or any link to external sites. The use of the website is at the user’s sole risk and responsibility. This information platform was created and maintained with the financial support of the European Union. Its contents do not, however, reflect the views of the European Union.
