Maximum levels for 3-MCPD in infant formulae
- Contaminants
- Food safety
Summary
The European Union (EU) has notified the World Trade Organization Sanitary and Phytosanitary Measures (WTO SPS) Committee of a new draft proposal to set or reduce maximum levels for glycidyl fatty acid esters (expressed as glycidol) and 3-monochlorpropanediol (3-MCPD) – sum of 3-MCPD and 3-MCPD fatty acid esters – in certain processed cereal-based foods for babies, infants, and young children, and in vegetable oils and fats, fish oils, and oils from other marine organisms used for the production of those foods (G/SPS/N/EU/935).
In January 2025, the EU reduced the maximum levels of the sum of 3-MCPD and its esters in certain specific food products for infants and young children (Regulation 2024/1003).
EU reduces glycidol and 3-MCPD maximum levels in infant formula and food
Draft Commission Regulation amending Regulation (EU) 2023/915 as regards maximum levels of 3‐monochloropropanediol (3-MCPD), 3-MCPD fatty acid esters and glycidyl fatty acid esters in certain foods
Draft Annex
Commission Regulation (EU) 2024/1003 of 4 April 2024 amending Regulation (EU) 2023/915 as regards maximum levels for the sum of 3-monochlorpropanediol (3-MCPD) and 3-MCPD fatty acid esters in infant formulae, follow-on formulae and food for special medical purposes intended for infants and young children and young child formulae
Update
The European Union (EU) has notified the World Trade Organization Sanitary and Phytosanitary Measures (WTO SPS) Committee of a new draft proposal to set or reduce maximum levels for glycidyl fatty acid esters (expressed as glycidol) and 3-monochlorpropanediol (3-MCPD) – sum of 3-MCPD and 3-MCPD fatty acid esters – in certain processed cereal-based foods for babies, infants, and young children, and in vegetable oils and fats, fish oils, and oils from other marine organisms used for the production of those foods (G/SPS/N/EU/935).
In January 2025, the EU reduced the maximum levels of the sum of 3-MCPD and its esters in certain specific food products for infants and young children (Regulation 2024/1003).
Impacted Products
Processed cereal-based food for infants and young children; compound foods containing more than 5% fat; vegetable oils and fats and/or oils from fish and/or other marine organisms added to baby food; processed cereal-based foods for infants and young children; infant formulae, follow-on formulae, and food for special medical purposes intended for babies, infants, and young children
What is changing?
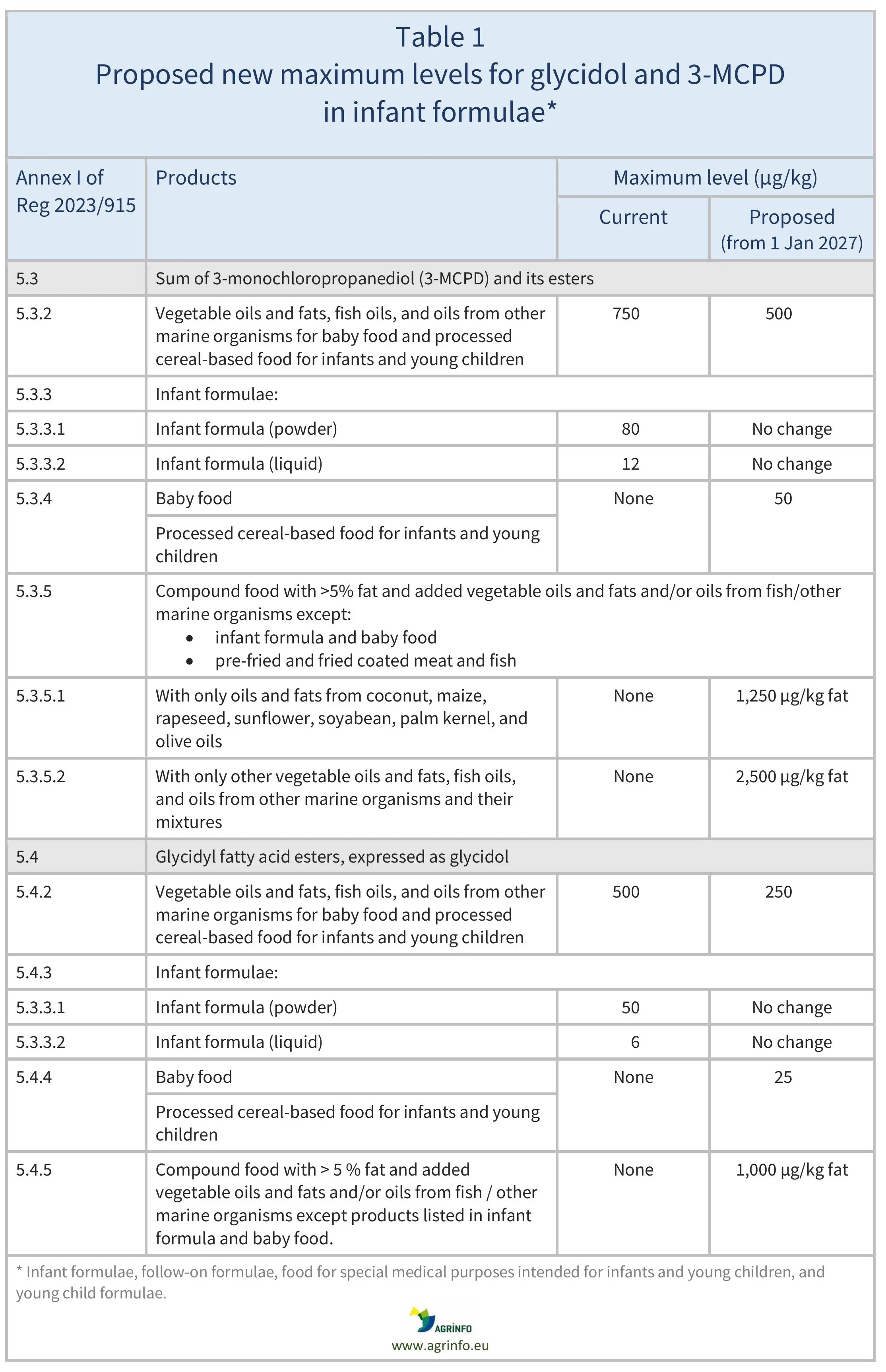
The European Commission proposes to set new maximum levels for glycidol and 3-MCPD in:
- baby food and processed cereal-based food for infants and young children
- compound food containing more than 5% fat, and added vegetable oils and fats and/or oils from fish and/or other marine organisms.
It also proposes reducing maximum levels of these contaminants in:
- vegetable oils and fats, fish oils, and oils from other marine organisms that are used in the production of baby food
- processed cereal-based food for infants and young children.
The proposed changes are summarised in Table 1.
Why?
The European Food Safety Authority (EFSA) has expressed health concerns about the presence of these substances in foods, especially in foods for infants and young children (EFSA 2016, 2018).
Timeline
The new maximum levels will apply from 1 January 2027.
Recommended Actions
Non-EU suppliers of these products for infants should evaluate current levels of glycidyl fatty acid esters (expressed as glycidol) and 3-MCPD and its esters in these foods to check compliance or consider strategies for reducing the presence of these contaminants.
Competent authorities of countries that are members of the WTO can submit comments on the EU’s proposal by emailing the EU SPS Enquiry Point until 26 May 2026.
Background
3-MCPD and glycidyl esters are contaminants of processed vegetable oils; free MCPDs are formed in some processed foods, particularly when high temperatures are applied to fats and oils.
Maximum levels for glycidol and for 3-MCPD were established in Regulation 2023/915 for:
- vegetable oils and fats
- fish oils and oils from other marine organisms
- infant formulae and follow-on formulae
- food for special medical purposes intended for infants and young children, and young child formulae.
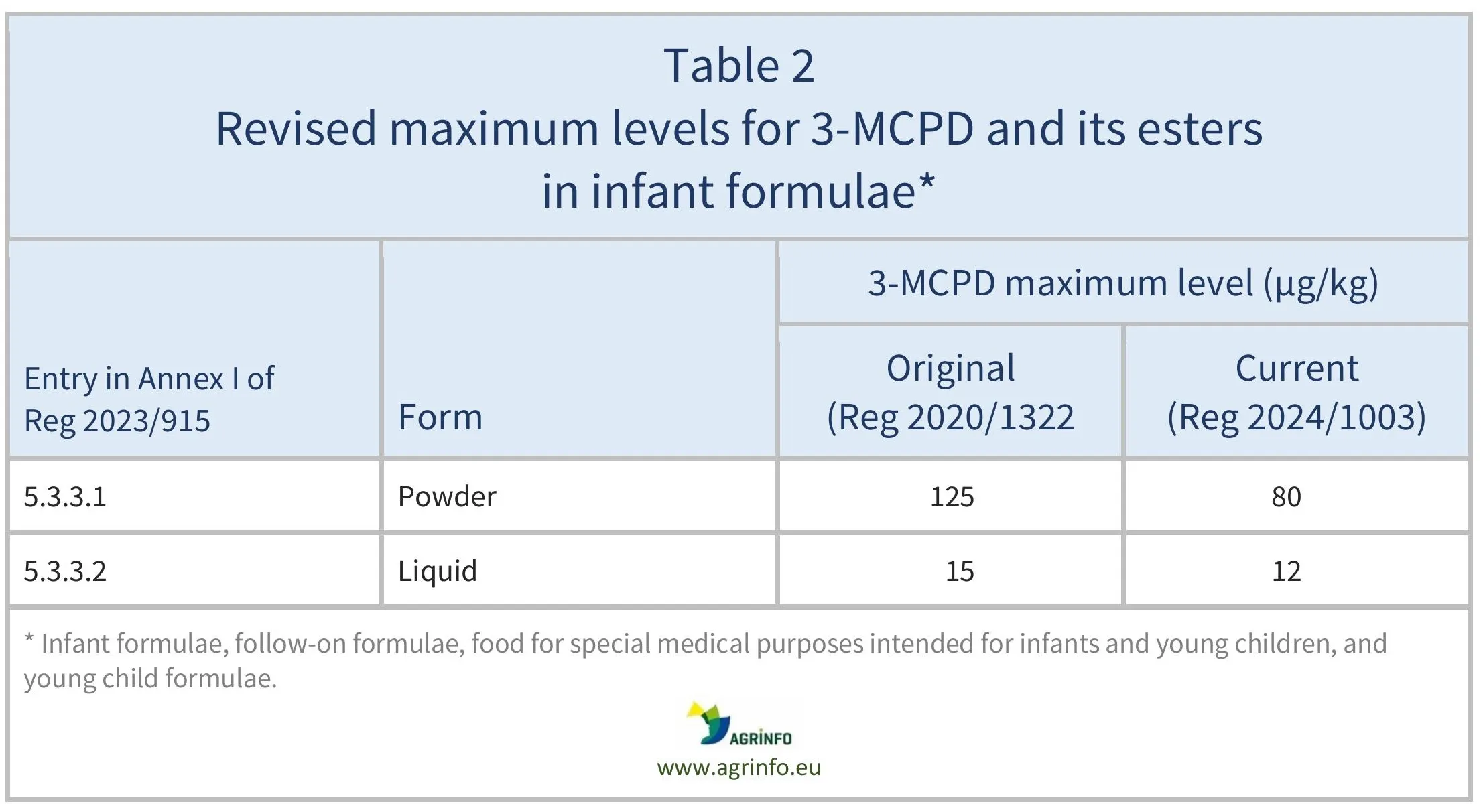
The original maximum levels for 3-MCPD and its esters in specific food products for infants and young children were established by Regulation 2020/1322.
Reduced maximum levels of the sum of 3-MCPD and its esters in infant formulae, follow-on formulae, food for special medical purposes intended for infants and young children, and young child formulae have applied since 1January 2025 (Regulation 2024/1003). These changes are shown in Table 2.
The EU aims to set maximum levels following the principle that they should be as low as reasonably achievable when applying good practices, and on the basis of scientific advice provided by EFSA, taking into account data on the occurrence of contaminants in foodstuffs from various origins. See EU legislation on contaminants – maximum levels explained.
Resources
EFSA (2016) Risks for human health related to the presence of 3- and 2-monochloropropanediol (MCPD), and their fatty acid esters, and glycidyl fatty acid esters in food. EFSA Journal, 14(5): e04426.
EFSA (2018) Update of the risk assessment on 3-monochloropropane diol and its fatty acid esters. EFSA Journal, 16(1): e05083.
European Commission (2008) Factsheet: Food contaminants.
Regulation (EU) 2024/1003 as regards maximum levels for the sum of 3-monochlorpropanediol (3-MCPD) and 3-MCPD fatty acid esters in infant formulae, follow-on formulae and food for special medical purposes intended for infants and young children and young child formulae
Regulation (EU) 2023/915 on maximum levels for certain contaminants in food
Regulation (EU) 2020/1322 as regards maximum levels of 3‐monochloropropanediol (3-MCPD), 3-MCPD fatty acid esters and glycidyl fatty acid esters in certain foods
Regulation (EEC) No 315/93 laying down Community procedures for contaminants in food
Sources
Draft Commission Regulation as regards maximum levels of 3‐monochloropropanediol (3-MCPD), 3-MCPD fatty acid esters and glycidyl fatty acid esters in certain foods
Draft Annex
Commission Regulation (EU) 2024/1003 as regards maximum levels for the sum of 3-monochlorpropanediol (3-MCPD) and 3-MCPD fatty acid esters in infant formulae, follow-on formulae and food for special medical purposes intended for infants and young children and young child formulae
Disclaimer: Under no circumstances shall COLEAD be liable for any loss, damage, liability or expense incurred or suffered that is claimed to have resulted from the use of information available on this website or any link to external sites. The use of the website is at the user’s sole risk and responsibility. This information platform was created and maintained with the financial support of the European Union. Its contents do not, however, reflect the views of the European Union.
EU reduces glycidol and 3-MCPD maximum levels in infant formula and food
Draft Commission Regulation as regards maximum levels of 3‐monochloropropanediol (3-MCPD), 3-MCPD fatty acid esters and glycidyl fatty acid esters in certain foods
Draft Annex
Commission Regulation (EU) 2024/1003 as regards maximum levels for the sum of 3-monochlorpropanediol (3-MCPD) and 3-MCPD fatty acid esters in infant formulae, follow-on formulae and food for special medical purposes intended for infants and young children and young child formulae
What is changing and why?
The European Commission proposes to set new maximum levels for glycidol and 3-MCPD in:
- baby food and processed cereal-based food for infants and young children
- compound food containing more than 5% fat, and added vegetable oils and fats and/or oils from fish and/or other marine organisms.
It also proposes reducing maximum levels of these contaminants in:
- vegetable oils and fats, fish oils, and oils from other marine organisms that are used in the production of baby food
- processed cereal-based food for infants and young children.
The proposed changes are summarised in Table 1.
The European Food Safety Agency has expressed health concerns about the presence of these substances, especially in foods for infants and young children.
Reduced maximum levels of the sum of 3-MCPD and its esters in infant formulae, follow-on formulae, food for special medical purposes intended for infants and young children, and young child formulae have applied since 1January 2025. Those changes are shown in Table 2.
Actions
Non-EU suppliers of these products for infants should evaluate current levels of glycidol and 3-MCPD in these foods to check compliance or consider strategies for reducing the presence of these contaminants.
Competent authorities of countries that are members of the World Trade Organization can submit comments on the EU’s proposal by emailing the EU SPS Enquiry Point until 26 May 2026.
Timeline
Applies from 1 January 2027.
Disclaimer: Under no circumstances shall COLEAD be liable for any loss, damage, liability or expense incurred or suffered that is claimed to have resulted from the use of information available on this website or any link to external sites. The use of the website is at the user’s sole risk and responsibility. This information platform was created and maintained with the financial support of the European Union. Its contents do not, however, reflect the views of the European Union.