French national MRL measures on carbendazim, thiophanate-methyl, glufosinate, and mancozeb
- Pesticide MRLs
Summary
On 7 January 2025, the French Government issued an Order suspending the import and sale of certain raw or processed foodstuffs, including some fruits, vegetables, soyabeans, cereals, and honey, that originate from outside the European Union (EU) if they contain carbendazim (sum of carbendazim and benomyl), thiophanate-methyl, glufosinate, or mancozeb. These pesticides are not approved for use in the EU.
The Order was notified to the World Trade Organization (WTO) on 9 January 2026, and has applied since 8 February 2026. It applies only to foods put on the French market, and not to foods put on other EU Member State markets.
The French Ministry of Agriculture has issued an FAQ document to help operators implement the new measures.
France bans imports of certain foods containing residues of carbendazim, thiophanate-methyl, glufosinate, and mancozeb
Arrêté du 5 janvier 2026 portant suspension d’importation, d’introduction et de mise sur le marché à titre gratuit ou onéreux, en France, de denrées alimentaires provenant de pays tiers à l’Union européenne contenant des résidus de certaines substances actives phytopharmaceutiques interdites d’utilisation dans l’Union européenne
Update
On 7 January 2025, the French Government issued an Order suspending the import and sale of certain raw or processed foodstuffs, including some fruits, vegetables, soyabeans, cereals, and honey, that originate from outside the European Union (EU) if they contain carbendazim (sum of carbendazim and benomyl), thiophanate-methyl, glufosinate, or mancozeb. These pesticides are not approved for use in the EU.
The Order was notified to the World Trade Organization (WTO) on 9 January 2026, and has applied since 8 February 2026. It applies only to foods put on the French market, and not to foods put on other EU Member State markets.
The French Ministry of Agriculture has issued an FAQ document to help operators implement the new measures.
Impacted Products
Apples, apricots, aubergines/eggplants, avocados, barley, beans (with pods), blackcurrants, Brussels sprouts, cherries (sweet), clementines/mandarins, grapefruit, grapes (wine and table), Japanese medlars, lemons, lettuce, limes, mangoes, medlars, melons, mushrooms (cultivated), oats, okra/lady’s fingers, oranges, papayas, peaches, pears, peas (with pods), sweet peppers/bell peppers, plums, pome fruits (other), potatoes, pumpkins, quinces, rye, seaweed and prokaryotic organisms, soyabeans, strawberries, tomatoes, watermelons, wheat
What is changing?
On 7 January 2025, the French Government issued an Order suspending the import and sale of some foodstuffs that originate from outside the EU if they contain certain pesticides that are not approved for use in the EU. The full decree in French is available at https://www.legifrance.gouv.fr/jorf/id/JORFTEXT000053313910.
This measure by the French authorities addresses the pesticides carbendazim (includes the sum of carbendazim and benomyl), thiophanate-methyl, glufosinate, and mancozeb, and their permitted MRLs on selected foods (raw or processed). The foods affected are listed in Table 1. In France, importing these foods and placing them on the market is now prohibited if they contain residues of any of these pesticides. This measure only applies to the French market; the newly established French MRLs are therefore not aligned with the EU MRLs currently in force across other EU Member States.
This Order entered into force on 8 January 2026, with a grace period of 1 month until it was applied, and was notified to the WTO on 9 January 2026. This very short transition period allowed little time for producers and traders to adapt to these new requirements.
On 20 January 2026, the European Commission and EU Member States discussed whether similar measures are required by the EU. The EU Member States did not support an EU-wide emergency measure, and will continue pursuing the following actions that are already planned:
- carbendazim, thiophanate-methyl, and benomyl: publication of a draft Regulation lowering maximum residue levels (MRLs) on all products (see AGRINFO records on carbendazim, thiophanate-methyl, and benomyl)
- glufosinate: update of risk assessment at EU level
- dithiocarbamates (including mancozeb): development of specific analytical methods.
Only one EU Member States supported repealing the French Order, and it was agreed that France may maintain its own emergency measure (European Commission 2026).
Why?
Under EU law, where an EU Member State informs the European Commission of an evident serious risk to human health, animal health, or the environment, and the Commission does not take measures to address that risk, a Member State may adopt interim protective measures (Regulation 178/2002, Arts. 53 and 54). This is the legal basis for the Order presented by France.
Timeline
This Order entered into force on 8 January 2026 and has been applied since 8 February 2026.
Background
Carbendazim (carbendazim + benomyl) and thiophanate-methyl
Carbendazim (carbendazim + benomyl) and thiophanate-methyl are no longer authorised for use in the EU as no application was made by the manufacturers for their reapproval. When substances are not reapproved, MRLs are set to the limit of quantification (LOQ), except on products for which an import tolerance is set based on uses outside the EU, and where these MRLs are considered safe by the European Food Safety Authority (EFSA). For carbendazim and thiophanate-methyl, EU import tolerances are currently in place on citrus fruits, mangoes, papayas, and okra/lady’s fingers.
In February 2026, the European Commission published adraft Regulation lowering maximum residue levels (MRLs) on all products (see AGRINFO records on carbendazim, thiophanate-methyl, and benomyl).
Glufosinate
Glufosinate has not been approved for use in the EU since July 2018 as no application was made by the manufacturer for its reapproval. It can still be used, but it should be substituted by safer alternatives where they exist.
Import tolerance MRLs currently remain in place for several products, of which only potatoes are included in the French Order: the MRL on potatoes was reduced to the LOQ, while for other foods the EU import tolerance MRL remains at 0.3 mg/kg (see the EU Pesticides Database).
Mancozeb
Mancozeb belongs to the dithiocarbamates group, which also includes maneb, metiram, propineb, thiram, and ziram. While these substances are not approved for use in the EU, several import tolerance MRLs remain in place.
In 2024, the European Commission informed the WTO that it intends to amend the MRLs for dithiocarbamates in a range of products (G/SPS/N/EU/788). For products where Codex MRLs (CXLs) or import tolerances exist and are considered safe, the Commission proposed adjusting the MRLs accordingly. For details on the proposed changes and products affected, see Maximum residue levels for dithiocarbamates.
As limited data is currently available for certain products, further evaluations and potential adjustments are still ongoing. Adoption of this proposal was originally planned for 2025 but is on hold pending further discussions within the Commission.
Resources
EFSA (2021) Reasoned opinion on the toxicological properties and maximum residue levels (MRLs) for the benzimidazole substances carbendazim and thiophanate-methyl. EFSA Journal, 19(7): e06773.
Regulation (EC) No 178/2002 laying down the general principles and requirements of food law, establishing the European Food Safety Authority and laying down procedures in matters of food safety.
European Commission (2026) Standing Committee on Plants, Animals, Food and Feed, Section Phytopharmaceuticals – Residues. Summary Report, 20 January.
Ministère de l’Agriculture, de l'Agro-alimentaire et de la Souveraineté alimentaire (2026) Foire aux questions : suspension de la mise sur le marché en France de denrées alimentaires provenant de pays tiers contenant des résidus de substances actives phytopharmaceutiques interdites dans l’Union européenne
Sources
Arrêté du 5 janvier 2026 portant suspension d’importation, d’introduction et de mise sur le marché à titre gratuit ou onéreux, en France, de denrées alimentaires provenant de pays tiers à l’Union européenne contenant des résidus de certaines substances actives phytopharmaceutiques interdites d’utilisation dans l’Union européenne
Tables & Figures
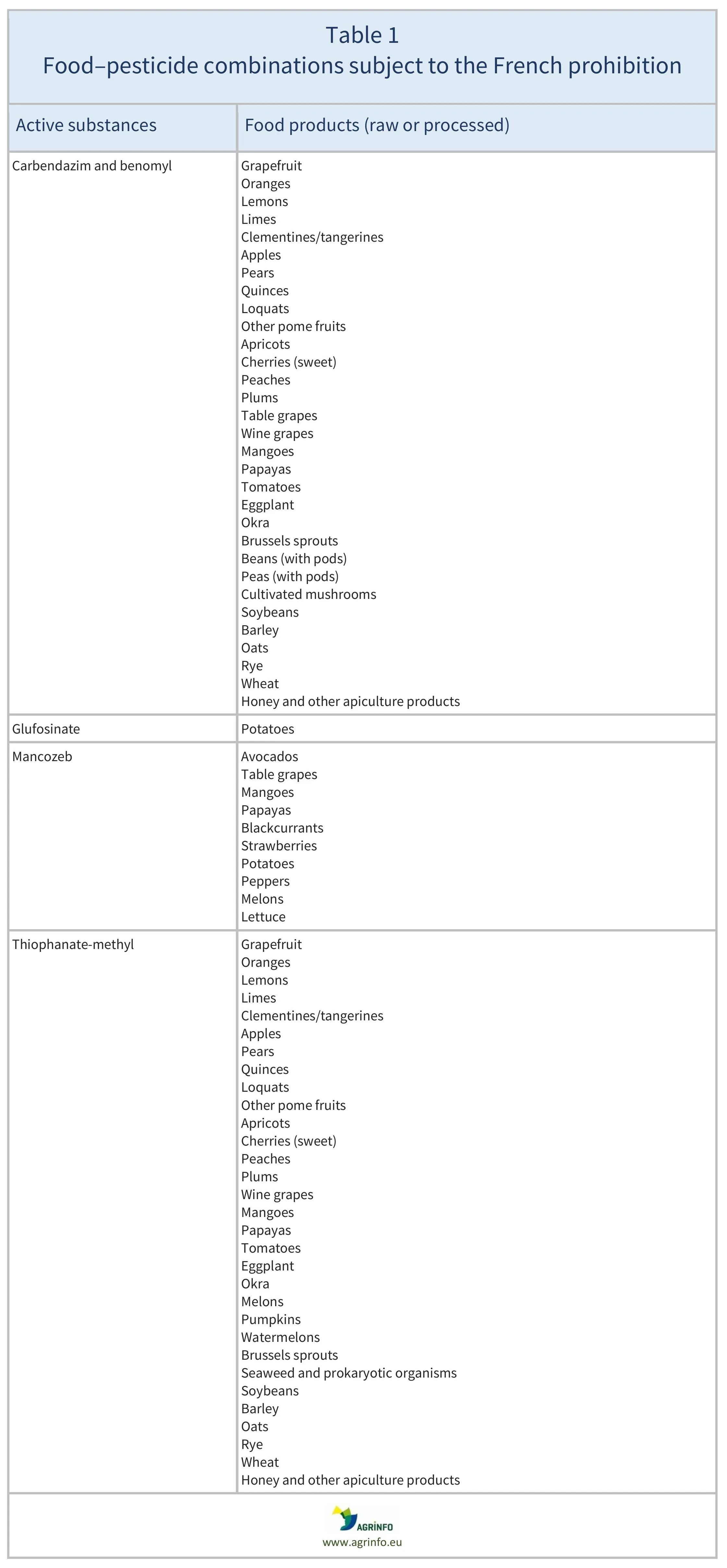
Source: based on Art. 1 of the French Order
Disclaimer: Under no circumstances shall COLEAD be liable for any loss, damage, liability or expense incurred or suffered that is claimed to have resulted from the use of information available on this website or any link to external sites. The use of the website is at the user’s sole risk and responsibility. This information platform was created and maintained with the financial support of the European Union. Its contents do not, however, reflect the views of the European Union.
France bans imports of certain foods containing residues of carbendazim, thiophanate-methyl, glufosinate, and mancozeb
Arrêté du 5 janvier 2026 portant suspension d’importation, d’introduction et de mise sur le marché à titre gratuit ou onéreux, en France, de denrées alimentaires provenant de pays tiers à l’Union européenne contenant des résidus de certaines substances actives phytopharmaceutiques interdites d’utilisation dans l’Union européenne
What is changing and why?
On 7 January 2025, the French Government issued an Order suspending the import and sale of some foodstuffs that originate from outside the European Union (EU) if they contain certain pesticides that are not approved for use in the EU. This measure by the French authorities addresses the pesticides carbendazim (includes the sum of carbendazim and benomyl), thiophanate-methyl, glufosinate, and mancozeb. The Order has applied since 8 February 2026. It applies only to foods put on the French market, and not to foods put on other EU Member State markets.
The French Ministry of Agriculture has issued an FAQ document to help operators implement the new measures.
Individual EU Member States are permitted under EU law to take national emergency measures only where there is an evident serious risk to human health. The Order was notified to the World Trade Organization on 9 January 2026.
On 20 January 2026, the European Commission and EU Member States discussed the French Order. The EU Member States did not support an EU-wide emergency measure like that proposed by France, and instead will continue to pursue actions that are already planned on these substances. The EU Member States did not support repealing the French Order, and it was agreed that France may maintain its own emergency measure.
Timeline
This Order has applied since 8 February 2026.
Tables & Figures

Source: based on Art. 1 of the French Order
Disclaimer: Under no circumstances shall COLEAD be liable for any loss, damage, liability or expense incurred or suffered that is claimed to have resulted from the use of information available on this website or any link to external sites. The use of the website is at the user’s sole risk and responsibility. This information platform was created and maintained with the financial support of the European Union. Its contents do not, however, reflect the views of the European Union.
